
- •Terrestrial and atmospheric radiation
- •Radiation of the atmosphere
- •Carbon dioxide
- •Ozone
- •The IR radiation transmission function P for cloudiness depends on the mass of
- •Equation of the terrestrial radiation transfer in the atmosphere
- •Reasons for the change
- •Temperature and IR radiation fluxes relation
- •Layer

Terrestrial and atmospheric radiation
Earth’s surface radiation
As well as any other body with temperature T>0K, the Earth radiates energy. Since the Earth’s temperature is rather low
(T=288K), it radiates energy in |
part of the spectrum. |
||||||||
Neither Earth, nor its atmosphere, is not absolutely black |
|||||||||
0,85...0,99 |
body. Therefore |
B0 |
T04 |
|
|
0,95 |
|||
|
|||||||||
B0 BSun |
however |
B0 and |
I! |
are of the same order |
|||||
|
|
|
|
|
of magnitude. |
|
|
|
|
T°C |
-40 |
-20 |
0 |
20 |
40 |
|
|
|
|
B kW/m² |
0,17 |
0,24 |
0,32 |
0,43 |
0,55 |
|
|
|
|
1

Radiation of the atmosphere
It is of more complex character. As it follows from Kirchhoff’s law, the energy is emitted by only those gases which absorb it.
H2O, CO2, O3 are the main absorbers. There are secondary absorbers (in IR area only): NO; N2O; N2O4; N2O5; C2H4; CH4.
Usually we neglect their activity in radiation process.
Water vapor
“WINDOW” of water vapor is 8,5…12 μ. For this wavelength interval the water vapor is most transparent (see table in the lecture note 5). This interval is often called window of minimal extinction.
The strongest water vapor absorption band fall on the spectrum interval 5,5..7,0 μ.
2
Carbon dioxide
Volume index of absorption by CO2 is presented below
, |
2,6 – 2,8 |
4,1 – 4,4 |
9,1 – 10,9 |
|
|
||||
, m 1 0,025 |
0,33 |
|
|
|
2 10 5 |
|
|||
12,9 – 17,1
0,83
|
|
This is the most |
Carbon dioxide absorbs |
|
important |
IR radiation very lightly |
|
absorption band |
in the atmospheric |
|
of the carbon |
window |
|
dioxide |
|
|
|
3

Ozone
Ozone has several absorption bands in the IR part of the spectrum. However, the only bands represents practical interest; that is around 9,65 μ band. It is situated just in the “window”. All other absorption bands of ozone are override by absorption of water vapor and carbon dioxide.
Transmission function for IR radiation total flux
The function below accounts for water vapor and carbon dioxide only.
P Qw.v. ,Qc.d. P1 Qw.v. P2 Qw.v. P3 Qc.d.
Mass of water vapor Qw.v.
Mass of carbon dioxide Qc.d.
1 m² |
1 m² |
|
4

|
3 |
|
|
|
3 |
|
P Qw.v. i exp i Qw.v. |
|
P Qc.d. i exp i Qc.d. |
||||
|
i 1 |
|
|
|
i 1 |
|
Functions |
P1 Qw.v. 10 |
P1 Qw.v. 10 100 |
P |
P |
||
1 |
|
|
|
|
2 |
3 |
0,19 |
|
0,19 |
0,05 |
0,32 |
||
2 |
0,21 |
|
0,26 |
0,19 |
0,40 |
|
3 |
0,35 |
|
0,29 |
-- |
0,28 |
|
1 |
47,0 |
|
43,0 |
0,29 |
0,056 |
|
|
|
|
|
|
|
|
2 |
1,1 |
|
1,0 |
0,023 |
0,0012 |
|
|
|
|
|
|
|
|
3 |
0,03 |
|
0,015 |
-- |
0 |
|
|
|
|
|
|
|
|
Cloud free atmosphere allows for passage of 17…35% of IR terrestrial |
|
radiation. Cloudiness makes on appreciable contribution into absorption |
|
of IR radiation. Mass absorption index of water droplets is 500 – 2000 |
|
cm²/g. That is much larger than for water vapor. |
5 |
The IR radiation transmission function P for cloudiness depends on the mass of water droplets containing in the vertical air column of a unit section (cloud water storage).
Qδ kg/m² |
0,01 |
0,03 |
0,07 |
P |
0,144 |
0,013 |
0,0004 |
Cloudiness practically absorbs all infrared radiation at Qδ >0,03. Real clouds always contain Qδ >0,03.
Due to strong absorption of the IR radiation by water vapor, carbon dioxide, and , particularly, by clouds, the significant part of the terrestrial radiation is absorbed by the atmosphere, while an appreciable part of the SR is allowed to pass through the atmosphere. Thus, the atmosphere provides for a strong warming effect of the Earth surface.
6

Equation of the terrestrial radiation transfer in the atmosphere
The basis for the reasoning is the energy brightness. Assuming isotropic field of radiation, relation between
flux of radiation (I) and energy brightness (J) will be |
Z |
||||
I J |
I J |
|
|
|
|
|
|
|
|
||
|
A′ |
θ |
|
||
|
|
|
|
||
Energy brightness coming |
|
|
|
||
from the upper hemisphere |
J |
|
|
|
|
|
A |
||||
|
|
|
|
||
Energy brightness coming |
J |
|
|
||
from the lower hemisphere |
|
|
|||
Z+dz
Z
At the point A |
J |
|
The change of the |
|
|||
|
|
|
|
At the point A’ |
J |
dJ |
brightness is dJ |
|
|
|
7 |
|
|
|

Reasons for the change
Absorption |
J adl |
dl AA' |
a is density of |
||
|
|
|
|
|
absorbing gases |
|
|
|
|
|
|
|
|
|
|
|
E is the brightness of |
Radiation of the layer dz |
E adl |
|
|||
|
BB radiation |
||||
Diffusion of the radiant energy
On one hand it is |
On the other hand it |
extinction of the |
results in increasing |
radiation |
of the brightness |
The sum effect of the diffusion is very small and can be neglected.
8
|
|
dJ |
J |
E adl |
dl |
|
dz |
|
|||||
dJ |
J E a |
dz |
|
|
cos |
||||||||
|
|
|
|||||||||||
|
|
|
|
dz |
|||||||||
|
|
|
|
||||||||||
cos |
|
Dividing by |
|||||||||||
|
|
cos |
|||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||
dJ |
|
|
|
|
|
|
equations of heat (IR |
||||||
|
|
|
|
|
|||||||||
dz |
cos E J a |
|
|||||||||||
|
|
|
|
|
|
|
|
These are differential |
|||||
|
|
From the similar |
|
|
|
radiation) transfer. |
|||||||
|
|
|
|
|
To obtain equations for |
||||||||
|
|
reasoning |
|
|
|
||||||||
|
|
|
|
|
upward (Uλ) and |
|
|
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|||
dJ |
cos J |
E a |
|
downward (Gλ) |
|
|
|
||||||
|
homogeneous fluxes, |
||||||||||||
|
|
these equations must be |
|||||||||||
dz |
|
|
|
|
|
|
|
integrated with respect |
|||||
|
|
|
|
|
|
|
|||||||
to θ from 0 to π/2 and with respect to azimuth from 0 to 2π
9

|
|
|
|
|
|
|
As result we get |
||||
|
U |
a B |
U |
|
|
G |
a G B |
||||
|
z |
|
|
z |
|||||||
|
|
|
|
|
|
|
|
|
|
||
|
|
B |
|
C 5 |
|
and |
1,66 |
||||
|
|
|
1 |
|
|||||||
Here |
|
|
|
|
|
|
|
|
|||
|
|
C2 |
1 |
|
|
|
|||||
|
|
|
|
|
|
|
|||||
|
|
|
|
exp |
|
|
|
|
|||
|
|
|
|
|
T |
|
|
|
|
|
|
Fluxes Uλ and Gλ satisfy to the following boundary conditions:
G 0 |
at |
Z |
U |
|
B 1 r G |
at |
Z 0 |
|||||
|
|
|
|
|
|
|
|
|
|
|
||
|
|
|
For practical purposes |
|
T |
|
1 Ф |
|||||
|
|
|
|
|
|
|
|
|
|
|||
|
U U d |
|
|
G G d |
|
|
|
z |
||||
|
|
|
|
t |
cp |
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
Some practical interest is related to vertical gradients of the quantities U, G, and Ф (effective flux ).
10
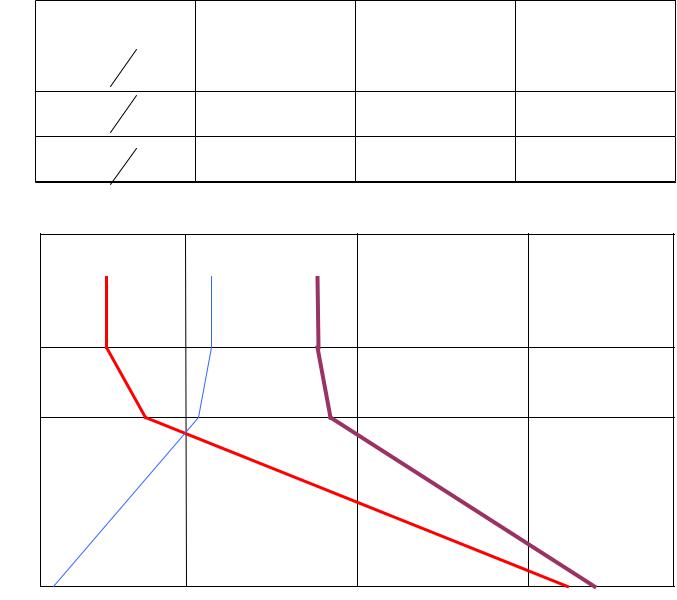
|
Levels |
P=P0 |
200 hPa |
100 hPa |
|
||
G |
kW |
m |
2 |
0,327 |
0,084 |
0,06 |
|
|
kW |
|
|
|
|
|
|
Ф |
m |
2 |
0,016 |
0,103 |
0,119 |
|
|
|
|
|
|
|
|
|
|
U |
kW |
m2 |
0,343 |
0,187 |
0,185 |
|
|
|
|
|
|
|
|
||
hPa |
|
|
|
|
|
|
|
|
G |
|
Ф |
|
U |
|
|
10 |
|
|
|
|
|
|
|
0 |
|
|
|
|
|
|
|
20 |
|
|
|
|
|
|
|
0 |
|
|
|
|
|
|
|
P=P0 |
|
|
|
|
|
|
|
|
|
|
0, 1 |
|
0, 2 |
0, 3 |
11 |
|
|
|
|
|
|
|
|
