
Color Atlas of Neurology
.pdf
Nonepileptic Seizures
Psychogenic seizure |
Eyes closed; patient squeezes |
(with arc de cercle) |
eyes shut when examiner |
|
attempts to open them |
Panic attack (hyperventilation, psychomotor restlessness)
Central Nervous System
203
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

Central Nervous System
204
Nonepileptic Seizures
! Drop Attacks
Sudden, unprovoked, and unheralded falls without loss of consciousness are most common in patients over 65 years of age. Some 10–15% of these drop attacks cause serious injury, particularly fractures. The patient may not be able to get up after a fall. The common causes of recurrent falls in each age group are listed in Table 26 (p. 374). Those associated with loss of consciousness are described on pages 192 ff and 200.
! Hyperventilation Syndrome (Tetany)
The clinical manifestations include paresthesiae (perioral, distal symmetrical or unilateral), generalized weakness, palpitations, tachycardia, dry mouth, dysphagia, dyspnea, yawning, pressure sensation in the chest, visual disturbances, tinnitus, dizziness, unsteady gait, muscle stiffness, and carpopedal spasms. The patients report feelings of restlessness, panic, unreality, or confusion. Psychological causes include anxiety, hysteria, and inner conflict. Metabolic causes include hypocalcemia (due to hypoparathyroidism, vitamin D deficiency, malabsorption, or pancreatitis) and a wide range of other disturbances including hypercalcemia, hypomagnesemia, prolonged vomiting, pulmonary embolism, salicylate intoxication, acute myocardial infarction, severe pain, high fever due to septicemia, pneumothorax, stroke, and neurogenic pulmonary edema. Chronic hyperventilation syndromes are more common than acute syndromes, but also more difficult to diagnose.
! Tonic Spasms
! Acute Dystonic Reaction
Acute dystonic reactions can occur within a few hours to one week of starting treatment with dopamine receptor antagonists, e. g., neuroleptics (benperidol, fluphenazine, haloperidol, triflupromazine, perphenazine), antiemetics (metoclopramide, bromopride), and calcium antagonists (flunarizine, cinnarizine). Symptoms and signs: focal or segmental dystonia (p. 64), sometimes painful, marked by oculogyric crisis, blepharospasm, pharyngospasm with glossospasm and laryngospasm, or oromandibular dystonia with tonic jaw and tongue movements. Generalized reactions are also seen on occasion (p. 66).
These are unilateral muscular spasms (often painful) that are not accompanied by loss of consciousness; they last seconds to minutes, and occur up to 30 or more times a day. They are most commonly seen in multiple sclerosis, less commonly in cerebrovascular disorders. These spasms are often triggered by movement. Some patients have paresthesiae (tingling, burning) contralateral to the affected side before the muscle spasm sets in. The underlying lesion may be in the brain stem (pons) or internal capsule.
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

Drop attack
Nonepileptic Seizures
Central Nervous System
Hyperventilation
Tonic spasms |
Acute dystonic reaction |
205 |
(muscular spasms on left) |
(oculogyric crisis, oromandibular/pharyngeal dystonia) |
|
|
|
|
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

Central Nervous System
206
Parkinson Disease: Clinical Features
The diagnosis of Parkinson disease (PD; sometimes termed idiopathic Parkinson disease, to distinguish it from symptomatic forms of parkinsonism, and from other primary forms) is mainly based on the typical neurological findings, their evolution over the course of the disease, and their responsiveness to levodopa (L- dopa). Longitudinal observation may be necessary before a definitive diagnosis of PD can be given. PD is characterized by a number of disturbances of motor function (cardinal manifestations) and by other accompanying manifestations of different kinds and variable severity.
Cardinal Manifestations
Bradykinesia, hypokinesia, and akinesia. Motor disturbances include slow initiation of movement (akinesia), sluggishness of movement (bradykinesia) and diminished spontaneous movement (hypokinesia); these terms are often used nearly interchangeably, as these disturbances all tend to occur together. Spontaneous fluctuations of mobility are not uncommon. The motor disturbances are often more pronounced on one side of the body, especially in the early stages of disease. They affect the craniofacial musculature to produce a masklike facies (hypomimia), defective mouth closure, reduced blinking, dysphagia, salivation (drooling), and speech that is diminished in volume (hypophonia), hoarse, poorly enunciated, and monotonous in pitch (dysarthrophonia). The patient may find it hard to initiate speech, or may repeat syllables; there may be an involuntary acceleration of speech toward the end of a sentence (festination). Postural changes include stooped posture, a mildly flexed and adducted posture of the arms, and postural instability. Gait disturbances appear in the early stages of disease and typically consist of a small-stepped gait, shuffling, and limping, with reduced arm swing. Difficulty initiating gait comes about in the later stages of disease, along with episodes of “freez- ing”—complete arrest of gait when the patient is confronted by doorway or a narrow path between pieces of furniture. It becomes difficult for the patient to stand up from a seated position, or to turn over in bed. Impairment of fine motor control impairs activities of daily living such as fastening buttons, writing (micro-
graphia), eating with knife and fork, shaving, and hair-combing. It becomes difficult to perform two activities simultaneously, such as walking and talking.
Tremor. Only about half of all PD patients have tremor early in the course of the disease; the rest usually develop it as the disease progresses. It is typically most pronounced in the hands (pill-rolling tremor) and is seen mainly when the affected limbs are at rest, improving or disappearing with voluntary movement. Its frequency is ca. 5 Hz, it is often asymmetrical, and it can be exacerbated by even mild stress (mental calculations, etc.).
Rigidity. Elevated muscle tone is felt by the patient as muscle tension or spasm and by the examiner as increased resistance to passive movement across the joints. Examination may reveal cogwheel rigidity, i.e., repeated, ratchetlike oscillations of resistance to passive movement across the wrist, elbow, or other joints, which may be brought out by alternating passive flexion and extension.
Postural instability (loss of balance). Propulsion and retropulsion arise in the early stages of Parkinson disease because of generalized impairment of the postural reflexes that maintain the bipedal stance. Related phenomena include involuntary acceleration of the gait (festination), difficulty in stopping walking, gait instability, and frequent falls.
Accompanying Manifestations
! Behavioral Changes
Depression. The range of depressive manifestations includes worry, anxiety, avoidance of social contact, general unhappiness, listlessness, querulousness, brooding, somatoform disturbances, and (rarely) suicidal ideation.
Anxiety. Tension, worry, mental agitation, lack of concentration, and dizziness are relatively common complaints.
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.
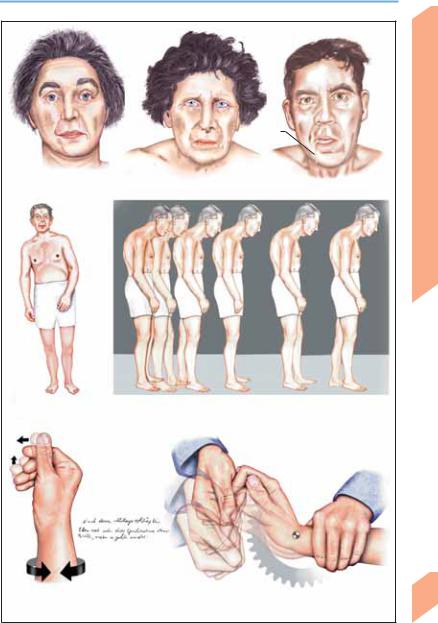
Parkinson Disease: Clinical Features
Hypomimia |
Drooling |
|
|
|
Facial expression |
Postural change |
Gait impairment (postural instability, propulsion, festination) |
|
(hypokinesia-left) |
||
|
Micrographia
Resting tremor |
Rigidity (cogwheel phenomenon) |
Central Nervous System
207
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

Parkinson Disease: Clinical Features
|
Dementia. Impairment of memory and concen- |
||||
|
tration in early PD-associated dementia may be |
||||
|
difficult to distinguish from depressive manife- |
||||
|
stations. The side effects of pharmacotherapy |
||||
|
(p. 212) must be kept in mind before treatment |
||||
|
is initiated for patients suffering from disorien- |
||||
|
tation, confusion, suspiciousness, and other |
||||
|
emotional changes. Impaired memory is usually |
||||
|
not a major feature of PD; as the disease pro- |
||||
|
gresses, about 20% of patients develop |
||||
|
decreased flexibility of thought and action, per- |
||||
System |
severation, and increasing difficulty in planning |
||||
future activities. The development of dementia |
|||||
in PD is correlated with an increase in the num- |
|||||
|
|||||
|
ber of Lewy bodies (pp. 210). |
|
|||
Nervous |
Hallucinations. A state of excessive suspicious- |
||||
ness, vivid dreams, and increasing anxiety may |
|||||
evolve into one of severe confusion with visual |
|||||
hallucinations. Frank psychosis (e. g., paranoid |
|||||
Central |
delusions, |
ideas of |
reference, or |
delusional |
|
jealousy) may be due to other causes than PD, |
|||||
|
|||||
|
particularly an adverse effect of antiparkin- |
||||
|
sonian medication. Dementia with Lewy bodies |
||||
|
(a syndrome in which the clinical features of |
||||
|
Parkinson disease are found together with |
||||
|
dementia, fluctuating level of consciousness, |
||||
|
visual hallucinations, and frequent falls) is |
||||
|
another possible cause, especially in patients |
||||
|
who are unusually sensitive to low doses of neu- |
||||
|
roleptics ( exacerbation of parkinsonism, |
||||
|
delirium, |
malignant |
neuroleptic |
syndrome, |
|
|
p. 347). |
|
|
|
|
! Autonomic Dysfunction
Blood pressure changes. Hypotension is a common side effect of antiparkinsonian medications (levodopa, dopamine agonists). Marked orthostatic hypotension, if present, suggests the possible diagnosis of multisystem atrophy.
Constipation may be caused by autonomic dysfunction, as a manifestation of the disease, or as a side effect of medication (anticholinergic agents).
Bladder disorders. Polyuria, urinary urgency, and urinary incontinence occur mainly at night and in patients with severe akinesia (who have difficulty getting to the toilet). PD only rarely causes severe bladder dysfunction.
208Sleep disorders. PD commonly causes disturbances of the sleep–wake cycle, including diffi-
culty falling asleep, nocturnal breathing prob-
lems similar to sleep apnea syndrome, and shortening of the sleep cycle. Sleep may also be interrupted by nocturnal akinesia, which makes it difficult for the patient to turn over in bed.
Sexual dysfunction. Spontaneous complaints of diminished libido or impotence are rare. Increased libido is a known side effect of levodopa and dopamine agonists.
Hyperhidrosis. Mainly occurs as generalized, irregular, sudden episodes of sweating. Seborrhea. Mainly on the forehead, nose, and scalp (greasy face, seborrheic dermatitis).
Leg edema is often the result of physical inactivity.
! Sensory Manifestations
Pain in the arm or shoulder, sometimes accompanied by fatigue and weakness, may be present for years before the cardinal manifestations arise and enable a diagnosis of PD. Back pain and nuchal cramps are frequent secondary effects of parkinsonian rigidity and abnormal posture. Dystonia may also come to attention because of the pain it produces.
Dysesthesia. Heat, burning or cold sensations may be felt in various parts of the body. For restless legs syndrome, see p. 114.
! Other Motor Manifestations
Dystonia. Tonic dorsiflexion of the big toe with extension or flexion of the other toes may occur in the early morning hours or during walking. Dystonia may be drug-induced (e. g., by levodopa) or due to the disease itself. The differential diagnosis includes dopa-responsive dystonia (a disorder of autosomal dominant inheritance) and Wilson disease (p. 307), two predominantly dystonic motor disorders with onset in childhood and adolescence.
Visual disturbances are caused by impairment of eye movement. Vertical gaze palsy is suggestive of progressive supranuclear palsy (p. 302). The reduced blinking rate of PD may lead to a burning sensation on the cornea, or to conjunctivitis.
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

Parkinson Disease: Clinical Features
Seborrhea
Orthostatic hypotension
Constipation
Urinary dysfunction, impotence
Behavioral changes |
Edema |
(depression, anxiety, dementia)
Autonomic dysfunction
Dystonia (of foot)
Sleep disorders
(increased rigidity at night, ”mental pillow”)
Pain
Central Nervous System
209
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

Parkinson Disease: Pathogenesis
|
Basal ganglia. The basal ganglia consist of the |
|
|
caudate nucleus (CN), putamen, globus pallidus |
|
|
(= pallidum; GPe = external segment, GPi = in- |
|
|
ternal segment; putamen + pallidum = lentiform |
|
|
nucleus), claustrum, substantia nigra (SN; SNc = |
|
|
pars compacta, SNr = pars reticularis), and the |
|
|
subthalamic nucleus (STN). CN + putamen = |
|
|
(dorsal) striatum; nucleus accumbens + portions |
|
|
of olfactory tubercle + anterior portion of puta- |
|
|
men + CN = limbic (ventral) striatum. Substantia |
|
|
nigra (SN): The SNr (ventral portion of SN) con- |
|
System |
tains small amounts of dopamine and iron, |
|
giving it a reddish color, while the SNc (dorsal |
||
portion) contains large quantities of dopamine |
||
|
||
|
and melanin, making it black (whence the |
|
Nervous |
name, substantia nigra). |
|
gated) neural pathways (circuits). Each circuit |
||
|
Connections. The basal ganglia are part of a |
|
|
number of parallel and largely distinct (segre- |
|
Central |
originates in a cortical area that is specialized |
|
for a specific function (skeletal motor, oculomo- |
||
|
||
|
tor, associative-cognitive, or emotional-moti- |
|
|
vational control), passes through several relay |
|
|
stations in the basal ganglia, and travels by way |
|
|
of the thalamus back to the cerebral cortex. Cor- |
|
|
tical projection fibers enter the basal ganglia at |
|
|
the striatum (input station) and exit from the |
|
|
GPi and SNr (output station). Input from the |
|
|
thalamus and brain stem also arrives at the stri- |
|
|
atum. Within the basal ganglia, there are two |
|
|
circuits subserving motor function, the so-called |
|
|
direct and indirect pathways. The direct path- |
|
|
way runs from the putamen to the GPi and SNr, |
|
|
while the indirect pathway takes the following |
|
|
trajectory: putamen GPe STN GPi SNr. |
|
|
The GPi and SNr project to the thalamus and |
|
|
brain stem. |
Neurotransmitters. Glutamate mediates excitatory impulses from the cortex, amygdala, and hippocampus to the striatum. Synapses from STN fibers onto cells of the GPi and SNr are also glutamatergic. Both the excitatory and the inhibitory projections of the SNc to the basal ganglia are dopaminergic. In the striatum, dopamine acts on neurons bearing D1 and D2 receptors, of which there are various subtypes (D1 group: d1, d5; D2 group: d2, d3, d4). D1 receptors predominate in the direct pathway, D2 receptors in the indirect path-
210way. Cholinergic interneurons in the striatum form a relay station within the basal ganglia
(transmitter: acetylcholine). Medium spiny-type
neurons (MSN) in the striatum have inhibitory projections to the GPe, GPi, and SNr (transmitters: GABA, substance P/SP, enkephalin/Enk). Other inhibitory GABAergic projections run from the GPi to the STN, from the GPi to the thalamus (ventrolateral and ventroanterior nucleus), and from the SN to the thalamus. The thalamocortical projections are excitatory.
Motor function. The direct pathway is activated by cortical and dopaminergic projections to the striatum. The projection from the striatum in turn inhibits the GPi, diminishing its inhibitory output to the thalamic nuclei (i.e., causing net thalamic activation). Thalamocortical drive thus facilitates movement initiated in the cerebral cortex (voluntary movement). In the indirect pathway, the striatum, under the influence of afferent cortical and dopaminergic projections, exerts an inhibitory effect on the GPe and STN. The result is a diminished excitatory influence of the STN on the GPi and SNr, ultimately leading to facilitation of cortically initiated voluntary movement and inhibition of involuntary movement.
! Pathophysiology
The cause of Parkinson disease is unknown. Its structural pathological correlate is a loss of neurons in the caudal and anterolateral parts of the SNc, with reactive gliosis and formation of Lewy bodies (eosinophilic intracytoplasmic inclusions in neurons) and Lewy neurites (abnormally phosphorylated neurofilaments) containing α- synuclein. Loss of pigment in the substantia nigra can be seen macroscopically. The most prominent neurochemical abnormality is a deficiency of dopamine in the striatum, whose extent is directly correlated with the severity of PD. The physiological effect of the lack of (mostly inhibitory) dopamine neurotransmission in the striatum is a relative increase in striatal activity, in turn causing functional disinhibition of the subthalamic nucleus via the indirect pathway. Meanwhile, in the direct pathway, decreased striatal inhibition of the GPi enhances the inhibitory influence of the GPi on the thalamus, leading to reduced activity in the thalamocortical projection. These changes in neural activity manifest themselves in the clinically observable akinesia, rigidity, and postural instability. For tremor, see p. 62.
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

|
|
|
|
|
Parkinson Disease: Pathogenesis |
|
|
Cerebral cortex |
|
Thalamocortical projections |
|
|
|
||
Thalamus |
|
|
|
|
|
|
|
CN |
|
|
|
Indirect |
|
Melanin |
|
|
|
|
pathway |
|
|
||
|
|
|
|
|
pigment |
|
|
|
|
|
|
Putamen |
|
|
|
|
|
|
|
|
|
|
|
STN |
|
|
|
GPe |
|
|
|
|
|
|
GPi |
|
|
System |
|
SN |
|
|
|
Neuron |
Lewy |
||
|
|
|
Nucleus |
||||
|
|
|
|
|
body |
|
|
|
|
|
|
accumbens |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Direct pathway |
|
|
Nervous |
|
|
|
Cerebral peduncle |
|
|
|
||
Superior |
|
|
Pons |
Thalamus |
Ventral lateral nucleus, ventral |
||
colliculus |
Red |
|
|
|
|
anterior nucleus |
Central |
|
nucleus |
|
|
|
|
(thalamus) |
|
|
|
|
|
|
|
||
Basal ganglia |
|
GL GL |
|
|
Putamen |
|
|
|
|
|
|
CN |
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
MSN |
|
|
|
ACh |
|
|
|
|
|
|
|
|
GABA,Enk GABA |
|
|
|
|
|
|
|
GABA |
|
|
|
|
|
|
|
GL |
|
|
|
|
|
|
DA |
|
|
GPi |
|
|
|
|
GABA,SP |
|
GPe |
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
STN |
|
|
|
|
|
|
GL |
|
|
|
|
|
|
|
|
GABA |
|
|
|
Direct pathway |
|
|
|
|
|
|
|
|
|
|
|
|
SNr |
|
|
Neurotransmitters: |
|
|
|
SNc |
|
||
GL: Glutamate |
|
|
|
|
|||
|
|
|
|
|
|
||
ACh: Acetylcholine |
|
|
|
|
Connections: |
|
|
DA: Dopamine |
|
|
|
|
|
||
|
|
|
|
|
|
Red: Excitatory |
|
|
|
|
|
|
|
Blue: Inhibitory |
|
|
|
|
|
|
|
Green: Excitatory and |
211 |
|
Functional organization (left, normal; right, Parkinson disease) |
inhibitory |
|
||||
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.

Central Nervous System
212
Parkinson Disease: Treatment
The goal of treatment is improvement of the motor, autonomic, and cognitive symptoms of the disease. The treatment generally consists of medication along with physical, occupational, and speech therapy. Neurosurgical procedures are mostly reserved for intractable cases (see below). Pharmacotherapy is palliative, not curative. It is begun when the patient has trouble carrying out the activities of daily living and is prescribed, not according to a uniform pattern, but in relation to the needs of the individual patient.
! Symptomatic Treatment
Dopaminergic agents. Levodopa is actively absorbed in the small intestine and rapidly distributed throughout the body (especially to skeletal muscle). Amino acids compete with the levodopa transport system at the blood–brain barrier. A decarboxylase inhibitor that does not penetrate the blood–brain barrier (benserazide or carbidopa) is administered together with levodopa to prevent its rapid breakdown in the peripheral circulation. Once it reaches the brain, levodopa is decarboxylated to dopamine, which is used for neurotransmission in the striatum. After it has been released from the presynaptic terminals of dopaminergic neurons in the striatum and exerted its effect on the postsynaptic terminals, it is broken down by two separate enzyme systems (deamination by monoamine oxidase type B, MAO-B; methylation by cate- chol-O-methyltransferase, COMT). Levodopa effectively reduces akinesia and rigidity, but has only a mild effect against tremor. Its long-term use is often complicated by motor fluctuations, dyskinesia, and psychiatric disturbances. Dopamine agonists (DAs) mimic the function of dopamine, binding to dopamine receptors. Their interaction with D1 and D2 receptors is thought to improve motor function, while their interaction with D3 receptors is thought to improve cognition, motivation, and emotion. Long-term use of DAs is less likely to cause unwanted motor side effects than long-term use of levodopa. Commonly used DAs include bromocriptine (mainly a D2 agonist), lisuride (mainly a D2 agonist), and pergolide (a D1, D2, and D3 agonist). Apomorphine, an effective D1 and D2 agonist, can be given by subcutaneous injection, but its effect lasts only about 1 hour. Other, recently in-
troduced dopamine agonists are ropinirol and pramipexol (D2 and D3), cabergoline (D2), and α- dihydroergocryptine (mainly D2).
Selegiline inhibits MAO-B selectively and irreversibly ( reduced dopamine catabolism increase in striatal dopamine concentration). Entacapone increases the bioavailability of levodopa via peripheral inhibition of COMT.
Nondopaminergic agents. Anticholinergic agents
(biperidene, bornaprine, metixene, trihexyphenidyl) act on striatal cholinergic interneurons. Budipine can relieve tremor (risk of ventricular tachycardia ECG monitoring). Glutamate antagonists (amantadine, memantine) counteract increased glutamatergic activity at the N-methyl-D-aspartate (NMDA) glutamate receptor in the indirect pathway.
! Transplant Surgery
Current research on intrastriatal transplantation of stem cells (derived from fetal tissue, from umbilical cord blood, or from bone marrow) seems promising.
!Stereotactic Neurosurgical Procedures, Deep Brain Stimulation (for abbreviations, see
p. 210)
These procedures can be used when PD becomes refractory to medical treatment. Pallidotomy (placement of a destructive lesion in the GPi) derives its rationale from the observed hyperactivity of this structure in PD. Deep brain stimulation requires bilateral placement of stimulating electrodes in the GPi or STN. Highfrequency stimulation by means of a subcutaneously implanted impulse generator can improve rigor, tremor, akinesia, and dyskinesia.
! Genetics of PD
A genetic predisposition for the development of PD has been postulated. Mutations in the genes for α-synuclein (AD), parkin (AR), and ubiquitin C-terminal hydrolase L1 (UCHL1; AD) have been found in pedigrees affected by the rare autosomal dominant (AD) and autosomal recessive (AR) familial forms of PD.
Rohkamm, Color Atlas of Neurology © 2004 Thieme
All rights reserved. Usage subject to terms and conditions of license.
