
The Diels-Alder Reaction
.pdf
M |
|
|
||
O |
O |
O |
O |
O |
|
||||
|
|
|
||
PhH, Rfx, 50% |
S |
AcOH, 43% |
||
|
|
|
73b |
|
O |
O |
|
|
|
O |
|
|
|
|
|
|
|
|
O |
|
|
O |
|
|
|
|
|
|
S |
|
|
|
|
S |
|
|
|
|
|
|
|
|
|
|
O |
|
|
|
|
+ |
80% |
O |
|
|
|
|
||
|
|
|
|
|
|
|
|
S |
|
|
S |
|
|
|
|
|
|
|
|
|
|
O |
O |
|
|
77 |
|
|
|
|
|
|
|
|
N |
N |
N |
N |
Ph |
Ph |
78 |
79 |
M

|
M |
MeO2C
O R
MA, PhH
Rfx, 24−60 h
80−82%
O O
O
O R
MP, PhMe, Rfx, 7 d,
12%
OMe
O R
80
R = H, Me
TCNE, CHCl3 r.t., 12−24 h
90%
NC CNCN
CN
MeO
O
BQ, PhMe, Rfx, 48 h, 32%
DMAD,
PhH or xylene Rfx, 5−72 h 25−33%
O
O R
MeO2C CO2Me
O R
O R
MP = methylpropiolate; BQ = 1,4-benzoquinone; MA = maleic anhydride; DMAD = dimethylacetylenedicarboxylate; TCNE = tetracyanoethylene
M
M
M

M |
|
|
R2 |
|
H O |
|
|
|
|
|
|
||
|
O |
H |
X |
|
|
|
|
|
|
||
|
R |
|
X = O, N-Ph |
|
|
|
|
1 O |
|
||
|
|
MA or NPM, |
|
||
|
|
PhH, r.t. |
|
|
|
|
|
3 h, 60−80% |
|
||
R2 |
|
R2 |
|
|
R2 |
H O |
NQ,n-PrOH |
|
|
TCNE, PhH, r.t. |
CN |
|
R1 |
CN |
|||
O |
r.t., 44% |
|
0.5−1 h, 50−83% |
CN |
|
|
|
O R1 CN |
|||
RH1 |
|
O |
|
|
|
O |
|
81 |
|
|
|
R1 = R2 = H, Me
MA = maleic anhydride; NPM = N-phenylmaleimide; NQ = 1,4-naphthoquinone; TCNE = tetracyanoethylene
O
O
S
BQ, AcOH,100 C 8 h, 51%
O |
|
|
|
|
|
CNCN |
|
O |
|
|
|
NC |
CN |
|
|
|
|
|
||
|
NQ, n-PrOH |
|
TCNE, THF, |
|
|
|
S |
r.t., 44% |
S |
0−25 |
C, 21 h,66% |
|
S |
|
|
|
|
|||
|
|
82 |
|
|
|
|
COPh
PhH, Rfx, 8 h, 54%
COPh
PhOC COPh
S
BQ = 1,4-benzoquinone; NQ = 1,4-naphthoquinone; TCNE = tetracyanoethylene

|
|
|
|
|
M |
|||
X |
Z |
X |
|
|
|
|
E |
E |
|
|
R1 |
|
|
||||
H |
R1 |
Z |
|
E |
E |
H |
R1 |
|
|
|
|
|
|||||
|
|
PhMe, MS 4A, |
N |
R |
|
PhMe, Rfx, |
|
|
N |
R |
Rfx, 7−40 h, |
H |
|
3−16 h, 20−46% |
N |
R |
|
H |
|
12−73% |
83 |
|
|
|
H |
|
|
|
|
|
|
|
|
|
|
|
|
|
R = H, Me; R1 = H, Me, Ph |
|
|
|
||
[1,3]-H shift |
|
|
|
|
|
[1,3]-H shift |
|
|
X |
Z |
|
|
|
|
|
− H2 |
|
|
R1 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N |
R |
|
|
|
|
|
E |
E |
|
|
|
|
|
|
|
||
H |
O Ph O |
|
|
|
|
|
|
R1 |
X-Z = |
; X = H, Z = CO2Me |
|
|
|
|
|||
N |
|
|
|
|
R |
|||
|
|
|
|
|
|
|
N |
|
X = H, Z = CO2Et; X = Z = CO2Me
H
E = CO2Me
$ $
|
CO2Me |
CO2Me |
|
CO2Me |
|
|
H |
CO2Me |
MeO2C |
|
|
|
||
N |
|
N |
|
N |
H |
|
H |
|
|
|
|
H |
||
|
|
|
|
|
H |
CO2Me |
|
|
! " |
PhMe, MS 4A |
|
CO2Me |
|
|
Rfx, 5 d, 8% |
|
− CH2=CH2 |
||
|
|
|
|
|
N |
|
N |
|
|
|
H |
CO2Me |
|
|
H |
|
|
||

M |
|
N N
84
M
R1 R2 |
|
|
|
R1 |
R2 |
|
|
|
|
CO2H |
acetone, Rfx |
|
CO2H |
HCl |
|
|
|||
|
|
|
|
|
|||||
N |
|
82−99% |
N |
CO2H |
Acetone |
r.t., |
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
84−96% |
|
|
|
|
O |
|
|
|
R1 R2 |
|
|
O |
|
|
|
|
|
|
H |
O |
|
|
|
|
H |
|
|
|
|
||
|
|
|
|
|
|
|
|
||
+ |
|
+ |
O |
|
|
|
|
|
|
|
|
|
|
|
|
O |
|||
R1 |
|
|
90−120 C, 3−18 h |
|
|
|
|||
|
|
|
|
|
|
||||
N |
R2 |
|
|
12−67% |
|
|
N |
H |
|
|
|
O |
|
|
|
||||
|
|
|
|
|
|
||||
|
|
|
|
|
|
|
O |
|
|
R2 |
|
|
|
|
R1 |
R2 |
|
|
|
|
|
|
|
H |
O |
|
|
||
R1 |
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
N |
H |
O |
|
|
|
|
|
|
|
|
|
|
||
|
|
|
|
|
H |
|
|
|
|
N |
|
|
|
|
|
O |
|
|
|
R1 = Me, Et, Ph, CH2COMe, p- and m-XC6H4 (X = Me, OMe, Br, Cl, F, NO, Ph) R2 = H, Me, Et, Bn, Me3CCOCH2

|
M |
M
#
R2 |
|
|
|
|
R2 |
|
R2 |
|
|
|
+ |
R* |
DCM, MS4A |
|
|
+ |
|
|
|
|
18−120 h, 20−40 |
|
|
|
|
|
|||
|
|
C |
|
R* |
|
|
R* |
||
N |
O |
|
N H |
N |
H |
||||
|
2−24% |
|
|
||||||
R1 |
|
|
|
R1 O |
|
R1 O |
|
||
|
|
|
|
|
exo |
|
endo |
|
|
|
|
|
|
|
R1 |
R2 |
exo/endo |
|
|
|
R* = |
N |
|
|
PhSO2 |
Me |
> 99:< 1 |
|
|
|
|
|
Me |
Me |
74:26 |
|
|
||
|
|
|
SO2 |
|
|
|
|||
|
|
|
|
Me |
Ph |
> 99:< 1 |
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
Me |
|
> 99:< 1 |
|
|
N
M
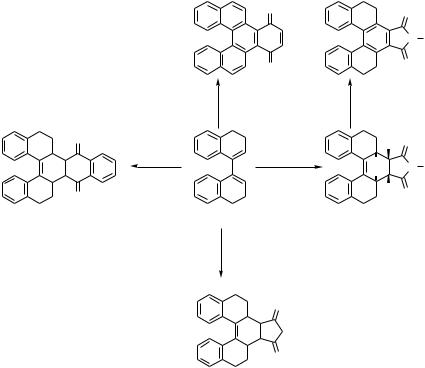
M |
|
O
O
BQ excess, 150 C, 22 h 25%
O
NQ |
NPM, 150 C |
50% |
3 h, 40% |
O
85
MA
NO2C6H5, 87%
O
O
O
N Ph
O
DDQ,triglyme, Rfx, 18 h 25%
H |
O |
|
H |
N Ph |
|
H |
||
|
||
H |
O |
BQ = 1,4-benzoquinone; NPM = N-phenylmaleimide; NQ = 1,4-naphthoquinone; MA = maleic anhydride

|
M |
1., DME
2.triglyme, Rfx, Pd/C, 30%
O |
|
|
O |
1. excess O |
, 150 C, 4.5 h |
2. triglyme, Rfx, Pd/C, 30% |
|
|
O |
|
O |
86 |
|
|
O |
1. excess |
O , PhMe, Rfx, 18 h |
2. triglyme, Rfx, Pd/C, 26%
O
M
M
M
M
M
M

M |
|
Ph |
|
|
|
Ph |
Ph |
|
+ |
|
r.t., −120 C |
|
+ |
|
R1 |
|
1−8 d, 40−92% |
|
|
|||
|
|
NC N R1 NC |
|
|
||
NC N |
R1 |
N |
! " |
|||
R |
|
|
|
R |
R |
|
|
|
|
|
|||
87 |
88 |
|
|
|
|
|
a, R = CO2Et |
R1 = Ph, EtO, CO2Me |
|
|
|
||
b, R = Ph |
|
|
|
|
|
|
c, R = OMe |
|
|
|
|
|
|
M
OH |
|
R |
R |
|
|
R |
Rfx |
|
C60 |
|
|
|
C60 |
|
|||
|
DCB |
|
|
||
NH |
N |
N |
! " |
||
|
|||||
|
|
|
|
89
R = Ph, 2-Th, p-MeOC6H4
|
R |
|
|
|
|
R |
|
|
|
N |
|
1. PhMe, 25 |
C, 6−24 h |
HN |
|
N |
|
|
+ |
R2CN |
|
|
|
|||
R1 |
|
|
|
|
|
|
||
N S |
|
2. H2O, 80−91% |
R1N |
S |
R2 |
|
||
|
|
|
|
! " |
||||
|
SiMe3 |
|
|
|
|
|
|
|
|
90 |
|
|
|
|
|
|
|
R = Ph, 2-Th; R1 = Ph, p -ClC6H4; R2 = Ts, CCl3, CO2Me

|
M |
|
|
|
O S |
|
|
O |
O |
|
|
R1 |
S |
|
R1 |
S |
|
|
|
|
|
|
|
||
|
|
|
O |
S |
O |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Py, CHCl3, |
|
|
|
|
|
Py, CHCl3, |
|
r.t., 24 h, |
|
|
|
|
|
r.t., 63−64 h, |
|
85% O |
|
|
|
|
|
79−89% |
|
, R1 = Me |
|
|
|
|
|
|
|
|
||
|
|
|
O |
O |
|
|
|
|
|
|
|
O |
|
|
|
|
|
|
|
|
|
O O |
|
|
O |
|
O |
|
S |
|
|
|
|
|
|
|
|||
R1 |
|
|
Py, CHCl3, r.t., |
R1 |
Py, CHCl3, r.t., |
R1 |
S |
S |
|
O |
|
||||
|
|
20−23 h, 88−97% |
91, R1 |
= Me 2.5−17 h, 67−78% |
O |
||
|
O |
|
|||||
|
|
|
92, R1 |
= OMe |
|
|
|
|
|
|
|
|
|
||
M
|
R |
|
|
R |
|
|
Py, CHCl3, r.t. R1 |
O O |
|
R1 |
O |
O |
+5−50 h, 58−87%
R2 |
S |
R2 |
|
S |
|
R3 |
! " |
||
|
R3 |
|
||
|
93 |
|
|
|
R = OH, OMe; R1 = Me, OMe; R-R1 = −(CH=CH)2−; R2-R3 = (CH=CH)2−; R-R1 and R2-R3 = −(CH=CH)2−
M
