
- •Course of lectures «Contemporary Physics: Part2»
- •The Wave Function
- •The Wave Function
- •The Wave Function
- •The Wave Function
- •The Wave Function
- •The Wave Function
- •The Wave Function
- •The Wave Function
- •SOLUTION
- •The Wave Function
- •The Wave Function
- •The Wave Function
- •Analysis Model: Quantum Particle
- •Analysis Model: Quantum Particle
- •Analysis Model: Quantum Particle A Particle in a Box Under Boundary Conditions
- •Analysis Model: Quantum Particle
- •Analysis Model: Quantum Particle A Particle in a Box Under Boundary Conditions
- •Analysis Model: Quantum Particle
- •Analysis Model: Quantum Particle
- •Analysis Model: Quantum Particle
- •The Schrödinger Equation
- •The Schrödinger Equation
- •The Schrödinger Equation
- •The Schrödinger Equation
- •The Schrödinger Equation
- •A Particle in a Well of Finite Height
- •A Particle in a Well of Finite Height
- •A Particle in a Well of Finite Height
- •A Particle in a Well of Finite Height
- •Tunneling Through a Potential Energy Barrier
- •Tunneling Through a Potential Energy Barrier
- •The Simple Harmonic Oscillator
- •The Simple Harmonic Oscillator
- •The Simple Harmonic Oscillator

Analysis Model: Quantum Particle
Under Boundary Conditions
Boundary Conditions on Particles in General
In general,
an interaction of a quantum particle with its environment represents one or more boundary conditions, and, if the interaction restricts the particle to a finite region of space, results in
 quantization of the energy of the system.
quantization of the energy of the system.

The Schrödinger Equation
Previously we discussed a wave equation for electromagnetic radiation that follows from Maxwell’s equations. The waves associated with particles also satisfy a wave equation. The wave equation for material particles is different from that associated with photons because material particles have a nonzero rest energy. The appropriate wave equation was developed by Schrödinger in 1926. In analyzing the behavior of a quantum system, the approach is to determine a solution to this equation and then apply the appropriate boundary conditions to the solution. This process yields the allowed wave functions and energy levels of the system under consideration. Proper manipulation of the wave function then enables one to calculate all measurable features of the system.

The Schrödinger Equation
The Schrödinger equation as it applies to a particle of mass m confined to moving along the x axis and interacting with its environment through a potential energy function U(x) is
where E is a constant equal to the total energy of the system (the particle and its environment). Because this equation is independent of time, it is commonly referred to as the time- independent Schrödinger equation. 

The Schrödinger Equation
The Particle in a Box Revisited
Because of the shape of the curve in Figure, the particle in a box is sometimes said to be in a square well, where a well is an upward- facing region of the curve in a potential-energy diagram.

The Schrödinger Equation
where A and B are constants that are determined by the boundary and normalization conditions.
everywhere, which is not a valid wave function

The Schrödinger Equation
Each value of the integer n corresponds to a quantized energy that we call En. Solving for the allowed energies En gives
Substituting the values of k in the wave function, the allowed wave functions are given by 

A Particle in a Well of Finite Height
In region II, where U=0
For regions I and III may be written
Because U>E, the coefficient of c on the right-hand side is necessarily positive. Therefore, we can express Equation as

A Particle in a Well of Finite Height

A Particle in a Well of Finite Height
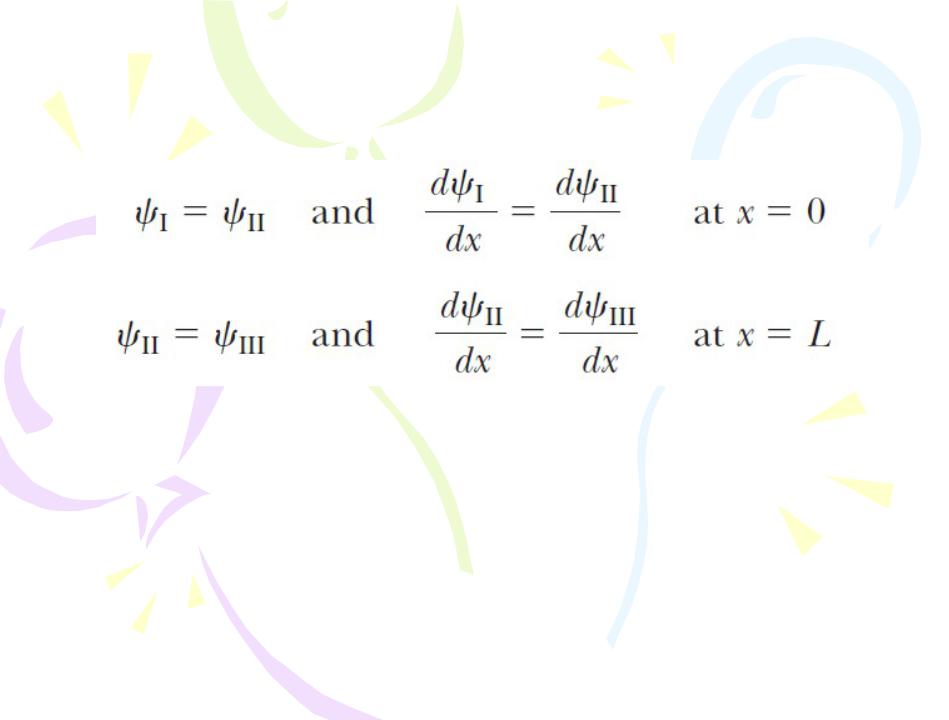
A Particle in a Well of Finite Height
In evaluating the complete wave function, we impose the following boundary conditions: 

