- •Section b gasoline (petrol) engines
- •Section c diesel engines
- •Section b water and its boiling point
- •C) molecules in motion
- •Section d engine-cooling system
- •Section b lubrication
- •Section c engine lubricating system
- •Section d filters
- •Unit four electricity Section a electrolysis
- •Uses of Electrolysis
- •Section b batteries
- •Section c automobile electric system
- •Section d battery-powered automobiles
- •Section b ferrous metals
- •Tensile strength and hardness section c
- •Section d aluminium bronze
- •Grammar exercises
- •Unit 1 advantages and disadvantages of diesel engines
- •Unit 2 comparative characteristics diesel and gasoline engines
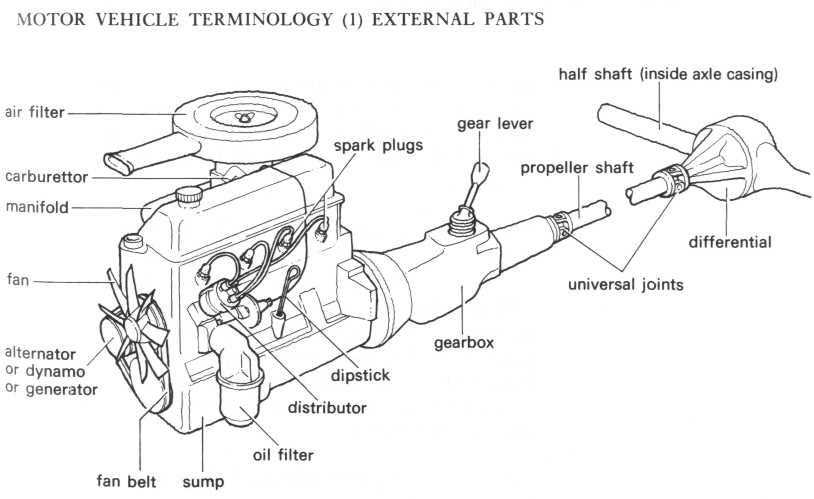
- •Unit 3 basic parts of the automobile
- •Unit 4 where diesel engines are used
- •Unit 5 Temperature Regulators of Cooling System
- •In machihes and machinery.
- •Unit 8 the instruments in a car
- •Unit 9 Battery chargers
- •Vocabulary Список сокращений
- •Literature used
- •Contents
C) molecules in motion
Look at the diagram about molecules in motion.

All matter is composed of molecules. Molecules are always in motion. Temperature affects the speed of the molecules.
For example, in a solid, each molecule remains in a stable position. However, it does not remain motionless. It vibrates rapidly between the adjacent molecules, but it does not escape from them. Therefore, a solid normally has stability, i.e. the surface, the shape and the volume are all stable. However, under certain conditions, a solid corrodes or decomposes. For example, certain metals corrode rapidly in acids and wood decomposes slowly in water.
A liquid does not have a stable shape, but it has a definite surface. Therefore it has a definite volume. In a liquid, each molecule moves rapidly among the other molecules. It does not remain in the same position, and a molecule sometimes breaks through the surface and escapes. At normal temperatures, a liquid evaporates slowly.
In a gas, each molecule moves at high speed. It does not remain among the other molecules. In an open container, it normally disperses rapidly into the atmosphere. It has no stability. It does not have a definite shape or a stable surface. Therefore, it does not have a stable volume either.
Exercise 7 Complete this table. |
|
|||
|
SOLIDS |
LIQUIDS |
GASES |
|
SURFACE |
|
definite but unstable |
indefinite and unstable |
|
SHAPE |
|
|
|
|
VOLUME |
|
|
|
|
Now make six sentences from this table.
A
In a |
solid liquid gas
solid liquid gas
|
each molecule
|
has does not have
remains does not remain
|
a definite shape. |
in a stable position.
|
Exercise 8 Add however or therefore to each pair of sentences.
Example: Water is normally a liquid.
Above 100°C, it becomes steam.
Water is normally a liquid. However, above 100°C, it becomes steam.
1. In a solid, each molecule remains in a stable position. It does not remain motionless.
2. In a solid, each molecule remains in a stable position. A solid has stability.
3. A solid normally has stability.
Under certain conditions, it corrodes or decomposes.
4. A liquid has a definite shape. It does not have a stable shape.
5. In a liquid, each molecule normally remains among the other molecules.
A liquid does not disperse into the atmosphere.
6. A liquid does not disperse into the atmosphere.
A molecule sometimes breaks through the surface and escapes.
7. A gas does not have a definite shape or a stable surface. It does not have a stable volume either.
8. A gas has no stability.
LANGUAGE NOTES
We may say in different ways. 1. Iron corrodes. Salt corrodes iron. 2. Wood decomposes. Water decomposes wood. 3. Ice melts. Heat melts ice. 4. Salt dissolves. Water dissolves salt.
We say: kemical, klorine but we write: chemical, chlorine
We say: sulfer, fysics, but we write: sulphur, physics.
Exercise 9
Check up whether you know the following words and expressions.
A compound, an atom, a molecule, a position, a surface, the atmosphere, chemical, (un)stable, (in)definite, rapid(ly), slow(ly), affect, remain, vibrate, escape break, move, disperse, evaporate, decompose, composed of, either, through,