
- •1.1 Introduction
- •1.2 Sample preparation and clean-up procedures
- •1.2.1 Liquid-liquid extraction
- •1.2.2 Solid phase extraction
- •1.2.3 Purge and trap
- •1.2.5 Derivatization
- •1.2.6 Clean-up procedures
- •1.3 Instrumentation
- •1.3.1 Gas chromatography
- •1.3.1.1 Capillary columns
- •1.3.1.2 Sample introduction systems
- •1.3.2 High performance liquid chromatography
- •1.3.2.1 Hplc columns
- •1.3.2.2 Hplc detectors
- •Volatile organic compounds
- •2.1 Introduction
- •2.2 Compounds
- •2.3 General Procedure
- •2.4 Sensitivity
- •3.1 Introduction
- •3.2 Compounds
- •3.3 General Procedure
- •3.4 Sensitivity
- •3.5.1 Procedure 1: Solid phase extraction/cgc/ms
- •4.1 Introduction
- •4.2 Compounds
- •4.3 General Procedure
- •4.4 Sensitivity
- •4.5 Detailed Procedures
- •4.5.1 Procedure 1: pah analysis using hplc (epa 550.0)
- •5.1 Introduction
- •5.2 Compounds
- •5.3 General Procedure
- •5.4 Sensitivity
- •6.1 Introduction
- •7.1 Introduction
- •7.2 Compounds
- •7.3 General Procedure
- •9.1 Introduction
- •9.3 General Procedure
- •9.4 Sensitivity
- •9.5 Detailed Procedures
- •9.5.1 Procedure 1: Tropolone extraction/cgc/aed or cgc/ms
- •10.1 Introduction
- •10.2 Compounds
- •10.3 General Procedure
- •10.4 Sensitivity
- •10.5 Detailed Procedures
- •11.1 Introduction
- •12.1 Introduction
- •12.2 Compounds
- •13.1 Introduction
- •100-90-80-70-60 50 40 30 20-10-0-
- •20 30 40
- •Iceland
- •Ireland
7.1 Introduction
The anilines and nitroaromatics of interest comprise some 36 compounds, seven of which are included in the EC priority pollutants list (EC). The determination of halogen and alkyl-substituted anilines and nitroaromatics in drinking, surface and ground water may be performed after liquid-liquid or solid phase extraction (SPE) by capillary gas chromatography with either nitrogen phosphorus (CGC/NPD) or mass spectroscopic (CGC/MS) detection. Special care must be taken when the presence of aniline precursors is suspected; urea herbicides for example, can generate anilines by thermal degradation in the sampling system.
A nilines
and nitroaromatics 7 163
nilines
and nitroaromatics 7 163
7.2 Compounds
Aniline
4-Bromoaniline
2-Chloroaniline (EC)
3-Chloroaniline (EC)
4-Chloroaniline (EC)
3 -Chloro- 4 -fluoraniline
3 -Chloro- 4 -methoxyaniline
-Chloro- 4 -methylaniline
-Chloro- 2 -methylaniline 2,4-Dichloroaniline (EC) 2,5-Dichloroaniline (EC) 2,6-Dichloroaniline (EC) 3,4-Dichloroaniline (EC) 2,6 -Diethylaniline
2,6 -Dimethylaniline 2 -Ethyl- 6 -methylaniline
Nitrobenzene
-Nitrotoluene
-Nitrotoluene 4-Nitrotoluene
.,3 -Dinitrotoluene 2,4-Dinitrotoluene 2,6-Dinitrotoluene
3.4 -Dinitrotoluene
2 -Methyl- 3 -nitroaniline 2 -Methyl- 4 -nitroaniline 2 -Methyl- 5 -nitroaniline 2 -Methyl- 6 -nitroaniline
-Methyl- 3 -nitroaniline
-Methyl- 2 -nitroaniline 2,4,6 -Trinitrotoluene
.,2 -Dinitrobenzene
.,3 -Dinitrobenzene
.,4 -Dinitrobenzene
2 -Amino- 4,6 -dinitrotoluene 4 -Amino- 4,6 -dinitrotoluene
(EC) Included on the European Community Priority Pollutants list.
 164
164
7 Anilines and nitroaromatios
7.3 General Procedure
Water is alkalized prior to solid phase extraction (pH9.0) or liquid-liquid extraction (pHll.O). Anilines and nitroaromatics are subsequently analysed by capillary gas chromatography/nitrogen phosphorus detection (CGC/NPD) or capillary gas chromatography/mass spectroscopy in single ion monitoring mode
(CGC/MS/SIM).
7.4 Sensitivity
0.025ppb for anilines. 0.050ppb for nitroaromatics.
7.5 Detailed Procedures
7.5.1 Procedure 1: Solid phase extraction/CGC/NPD or CGC/MS
Materials
• Chemicals
aniline standards
nitroaromatic standards
ethyl acetate, nanograde
heptadecanoic acid nitrile, internal standard
octadecanoic acid nitrile, internal standard
silica gel RP-C18
sodium hydroxide, p.a.
sodium chloride, p.a.
water, HPLC-grade
acetone, nanograde
methanol, nanograde
A nilines
and nitroaromatics7 165
nilines
and nitroaromatics7 165
Benzidines
8.1 Introduction
Benzidine and 3,3-dichlorobenzidine are poisonous, carcinogenic compounds formed as intermediates during the synthesis of dyes. Protective legislation limits their dispersal into the environment and both compounds are included on the EPA and EEC priority pollutants lists.
A quantitative analysis for water samples using HPLC with electrochemical detection is described.
8.2 Compounds
Compound names and structures are listed in Figure 8.1.
NH2
Benzidine
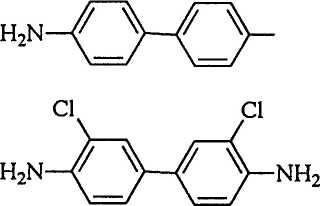
Figure 8.1: Chemical structures.
 Benzidines
8
Benzidines
8
181
8.3 General Procedure
Water at neutral pH is extracted using chloroform. The benzidines are back-extracted with 1M sulphuric acid and after neutralization, re-extracted with chloroform. Analysis is done by HPLC with electrochemical detection.
8.4 Sensitivity
O.Olppb
8.5 Detailed Procedure
8.5.1 Liquid-liquid extraction/HPLC/EC (EPA 605)
Materials
• Chemicals
benzidine, 3,3-dichlorobenzidine
methanol, nanograde
chloroform, nanograde
acetic acid, p.a.
sulphuric acid, p.a.
sodium hydroxide, p.a.
sodium acetate trihydrate
sodium phosphate, tribasic decahydrate
water, HPLC grade
acetonitrile, HPLC grade
• Solutions
• 5M sodium hydroxide
1M sulphuric acid
0.4MNa3PO4.10H2O
0.1M sodium acetate buffer : 5.8ml acetic acid and 13.6ml sodium acetate trihydrate in llitre HPLC water, Na2EDTA made up to O.OOIM.
• Tools
• 21 and 250 та\ sepaxatvm ^wwweYs
• rotary evaporator
HPLC instrument (isocratic) with amperometric detector
an HPLC column that can resolve the compounds shown in Figure 8.1
1 82 8
Benzidines
82 8
Benzidines
Organotin Compounds
