
- •1.1 Introduction
- •1.2 Sample preparation and clean-up procedures
- •1.2.1 Liquid-liquid extraction
- •1.2.2 Solid phase extraction
- •1.2.3 Purge and trap
- •1.2.5 Derivatization
- •1.2.6 Clean-up procedures
- •1.3 Instrumentation
- •1.3.1 Gas chromatography
- •1.3.1.1 Capillary columns
- •1.3.1.2 Sample introduction systems
- •1.3.2 High performance liquid chromatography
- •1.3.2.1 Hplc columns
- •1.3.2.2 Hplc detectors
- •Volatile organic compounds
- •2.1 Introduction
- •2.2 Compounds
- •2.3 General Procedure
- •2.4 Sensitivity
- •3.1 Introduction
- •3.2 Compounds
- •3.3 General Procedure
- •3.4 Sensitivity
- •3.5.1 Procedure 1: Solid phase extraction/cgc/ms
- •4.1 Introduction
- •4.2 Compounds
- •4.3 General Procedure
- •4.4 Sensitivity
- •4.5 Detailed Procedures
- •4.5.1 Procedure 1: pah analysis using hplc (epa 550.0)
- •5.1 Introduction
- •5.2 Compounds
- •5.3 General Procedure
- •5.4 Sensitivity
- •6.1 Introduction
- •7.1 Introduction
- •7.2 Compounds
- •7.3 General Procedure
- •9.1 Introduction
- •9.3 General Procedure
- •9.4 Sensitivity
- •9.5 Detailed Procedures
- •9.5.1 Procedure 1: Tropolone extraction/cgc/aed or cgc/ms
- •10.1 Introduction
- •10.2 Compounds
- •10.3 General Procedure
- •10.4 Sensitivity
- •10.5 Detailed Procedures
- •11.1 Introduction
- •12.1 Introduction
- •12.2 Compounds
- •13.1 Introduction
- •100-90-80-70-60 50 40 30 20-10-0-
- •20 30 40
- •Iceland
- •Ireland
12.2 Compounds
Names and structures of compounds that can be monitored with the described analytical techniques are given below.
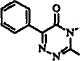
— CHj-CH
a \н,
\H,
ыи—сссн,»,
SCH,
CH,
<h,q,-
DesisopropylatrazineMetamitron
5 Desethylatrazine 9 Hexazinone
Simazine
Metribuzin
Cyanazine IS Atrazine
Oft-'
H f
H)C\
н,с
X
CjHj -
HC— HfT*N NH—С
Sebuthylazine
Propazine
н,с/
C,H,-
h,cn
HC-
н,с'
«3
. Jch 24 Terbuthylazine
P CH,
27 Prometryn
CH,
N N
SCH,
29 Terbutryn
C,H,-HN
..NH,
Other compounds that can be analyzed by the same method:
N^N CHi
11 Г
Fenuron 18 Isoproturon
Chloridazone 19 Metobromuron 6 Crimidin 20 Propham
NH-C<N
a ch,
8 Bromacil 21 Meuzachlor
Methabenzthiazuion 25 Iinuron
Chlortoluron 26 Chloroxuron
Monolinuron 28 Chlorprohara
Diuron 30 Metolachlor
 rfriazine
herbicides 12
rfriazine
herbicides 12
231
12.3 General Procedure
After solid phase extraction (SPE) of the water sample, triazines are analysed by high pressure liquid chromatography (HPLC) with UV detection, capillary gas chromatography/mass spectroscopy (CGC/MS) or capillary gas chromatography with nitrogen phosphorus detection (CGC/NPD).
12.4 Sensitivity
40 ppt in routine analysis.
12.5 Detailed Procedures
12.5.1 Procedure 1: Solid phase extraction/HPLC/DAD
Materials
• Chemicals
triazine reference compounds
methanol, p.a.
acetonitrile, p.a.
ethyl acetate, p.a.
water, HPLC-grade
• Solutions
• triazine stock solutions in methanol (lg/1)
• Tools
glass fibre filter paper
0.45 цт microfilters
solid phase extraction cartridges containing lg of octadecyl silica
solid phase extraction cartridges containing 500 mg of silica
SPE vacuum system
water-jet pump
blow-drying equipment
• Instrumentation
• liquid chromatograph with UV absorbance detector
. ODS HPLC column
2 32 12
Triazine herbicides
32 12
Triazine herbicides
Chloral hydrate
13.1 Introduction
Chloral hydrate is a polar chlorinated substance used in the manufacturing of DDT. Due to its toxicity it is included in the EC priority pollutants list.
13.2 Compounds
Chloral hydrate
CI3C CH OH
OH 2,2,2-trichloro-l, 1-ethanediol
13.3 General Procedure
Water samples are adjusted to pH13-14. Under alkaline conditions, the analyte is converted to chloroform, which can be analysed by purge and trap - capillary gas chromatography/mass spectrometry (CGC/MS).
13.4 Sensitivity
5-10ppb
C hloral
hydrate 13 243
hloral
hydrate 13 243
Trends and concerns
The most important developments in capillary GC and HPLC have been made within the last 10 years and the technological development of both techniques, based on known but as yet unexploited features, is proceeding unabated. Most notable are high speed capillary GC on ultra narrow bore columns, large volume injection in capillary GC, capillary GC/time-of-flight mass spectroscopy, electrospray micropacked LC/mass spectroscopy, LC/electrospray-collisional induced dissociation mass spectroscopy, an alternative to MS/MS and various multidimensional techniques including capillary GC/capillary GC, LC/capillary GC and LC/LC.
In Figure 14.1, the analysis of solvents on a 12.5m x 50|im i.d. capillary column with a 1 |xm film of methylsilicone demonstrates the current state-of-the-art in capillary GC.
This combination of high analysis speed and detectability, high resolution and an ability to accept conventional capillary column sample loads is waiting for the development of sample introduction systems suitable for routine analysis.
In a recent publication (1) the analysis of polychlorodibenzodioxins and polychlorobenzofurans was performed on a novel multidimensional CGC/CGC/MS system based on flow controlled column switching that allows the simultaneous isomeric separation of tetrachloro-, pentachloro- and hexachloro-substituted dibenzodioxins and dibenzofurans on a polar column and the analysis of heptachloro- and octachloro-substituted dibenzodioxins and dibenzofurans on an apolar column. Temperature programmed injection performed in the solvent venting mode allows the injection of sample volumes large enough to bring the sensitivity up to typical CGC/high resolution MS levels. On-line LC/CGC will provide environmental analysts with many new possibilities for sample preparation and trace enrichment, interested readers are referred to the excellent book on the subject by K. Grob (2).
Supercritical fluids have several characteristics which make them useful both as mobile phases for chromatography and for the selective extraction of organic components from different matrices. Far superior in terms of analysis times and
F. David, P. Sandra, A. Hoffmann and J. Gerstel, Chromatographia, 34 (1992), p. 259.
K.Grob, "On-Line Coupled LC-GC", Huthig Verlag GmbH, Heidelberg (1991)
T rends
and concerns 14 249
rends
and concerns 14 249
efficiency as compared to HPLC, the recent introduction of coupled packed columns and reliable instrumentation has moved SFC into the realm of routine analysis. Moreover, exotic detectors are not required, the compounds separated by SFC can be detected using standard GC detectors such as NPD and ECD. The 6 minute separation of sixteen priority pollutant PAHs shown in Figure 14.2 and the high efficiency separation of herbicides and pesticides using coupled columns shown in Figure 14.3 demonstrate the current state-of-the-art in SFC.
16 11
3-12 14
17
18 20
22
24 2»
26
29
30
31
32
13
3.3


7min
Figure 14.1: Separation of a solvents mixture by capillary gas chromatography. Peaks:
methanolethanol
aceton
diethylether
ethylformiate
dichloromethane
n-propanol
carbondisulfide
trans 1,2-dichloro- ethylene
methylethylketone
ethylacetate
cis 1,2-dichloroethylene
chloroform
tetrahydrofurane
n-butanol
1,2-dichloroethane (17)benzene
isooctane
p-dioxane
isobutylmethyl- ketone
(21)toluene
ethylpropylketone
dimethylformamide
butylacetate
n-butylether + m-xylene
p-xylene
cyclohexanol
o-xylene
dimethylmalonate + cyclohexanone
anisol
fenetol
benzylalcohol
dibutyl ketone
 250
250
14 Trends and concerns
![]()
