
Course Syllabus |
Module 1 |
Module 2 |
Module 3 |
Module Exams |
Final Exam |
Labs |
Quiestions & tasks |
handbook |
|||||||||||
Periodic table |
s-elements |
p-elements |
d-elements |
Fotogallery |
Video |
|
|
|
|||||||||||
[He]2s22p3 P [Ne] 3s23p3 As [Ar] 4s24p3 Sb [Kr] 5s25p3 Bi [Xe] 6s26p3
|
Halogens |
||||||||||||||||||
labs |
Home tasks |
Test problems |
Video |
photos |
|||||||||||||||
Properties of Halogens |
|||||||||||||||||||
Chemical |
Physical |
|
|||||||||||||||||
Uses |
Occurrence |
Preparation |
|||||||||||||||||
History Of Discovery |
|||||||||||||||||||
|
Electrode potentials |
||||||||||||||||||
Oxidation States |
Compounds of Halogens. Chemical and Physical Properties Overview |
||||||||||||||||||
N |
P |
As |
Sb |
Bi |
|||||||||||||||
-3
|
NH3 |
PH3 |
AsH3 |
SbH3 |
BiH3 |
||||||||||||||
NH4+(aq) |
PH4+(aq) |
|
|
|
|||||||||||||||
Li3N |
|
|
|
|
|||||||||||||||
BN |
|
|
|
|
|||||||||||||||
|
|
|
|
|
|||||||||||||||
-2 |
N2H4 |
P2H4 |
|
|
|
||||||||||||||
-1 |
NH2OH |
|
|
|
|
||||||||||||||
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|||||||||||||||
0 |
N2 |
P(red),P(white), P(black) |
As(grey), As (black) |
Sb(yellow) |
Bi |
||||||||||||||
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|||||||||||||||
+1 |
N2O |
H3PO2 |
|
|
|
||||||||||||||
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|||||||||||||||
+2 |
NO |
|
|
|
|
||||||||||||||
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|||||||||||||||
|
|
|
|
|
|||||||||||||||
+3 |
N2O3 |
P4O6 H3PO3 |
As2O3 H3AsO3 |
Sb2O3 |
Bi2O3 Bi(OH)3 |
||||||||||||||
+4 |
NO2 N2O4 |
|
|
|
|
||||||||||||||
+5 |
N2O5 |
P4O10 |
As2O5 |
Sb2O5 |
Bi2O5 |
||||||||||||||
HNO3 |
HPO3 |
H3AsO4 |
H3SbO3 |
HBiO3 |
|||||||||||||||
NO3-(aq) |
H4P2O7 |
HAsO3 |
K[Sb(OH)6] |
|
|||||||||||||||
|
H3PO4 |
|
|
|
|||||||||||||||
|
POCl3 |
|
|
|
|||||||||||||||
Electron configurations of nitrogen subgroup
Nitrogen
Nitrogen with four valence orbitals can be in the states of sp3-, sp2- and sp-hybridization like other second period elements|. There is a possibility of one or two -bonds (together with - bonds) formation in case of sp2- and sp- states,|formation| respectively. |figure,state,camp,mill|
The formation of nitrogen molecule is accompanied with release of significant amount of heat:
2N = N2 + 940 kJ/mol
The molecule of N2 is so extraordinarity stable that a lot of energy is required for its destruction. Additionally, internuclear distance is very short (0.1094 nm|) since the triple NN bond forms in the molecule. |curiously|The bond energy (940 kJ/mol) is equivalent to six single N—N bonds|truss| (160 kJ/mol), while|where as| the energy of triple СС bond |in the similar compound (С2Н2, 830 kJ/mol) is equivalent only to 2.5 single C—C bonds|truss| |. Whereas, a single N—N bond is considerably weaker (160 kJ/mol) than a single С—С bond (345 kJ/mol). Such a decrease in the strength of single homoatomic| bond for these elements is explained by the presence of antibonding| electrons in the atom of nitrogen increasing|aggravates| interatomic| repulsion. Therefore, chains —N—N—N— are so unstable unlike carbon chains.
The strongest in N2 molecule is the first bond (510 kJ/mol) that prevents|prevent| destruction of triple NN bond (the |changing|order of bond strength change in the series —С—С—, —С=С— and —СС— is|truss| contrary|return|). The energy diagram|chart| of N2 molecule is shown below:
2No = 2[1s22s22p3] = N2[KK(bonding2s)2(*2s)2(bonding2p)4(bonding2p)2]
The energy difference between s- and р-orbitals| is increased across the period towards |b group VIII elements. In accordance with this rule energies of 2s- and 2р-orbitals are nearly identical but electrons on 2s- and 2р- orbitals| have mutual repulsion, therefore bonding2рх and bonding2рy orbitals| become|stands| more favorable in energy and they are filled first,|earlier| not bonding2рz. Out of ten electrons of N2 molecule eight are located on bonding|coupling| orbitals|, and two - on antibonding ones. Six bonding|coupling| electrons are in excess|overabundance|, forming triple bond in N2.
Triple covalent| bond in N2 is absolutely|quite| unique. There are no other examples of diatomic homoatomic| molecules where bond of similar|like| type could be realized and could become so strong. From the point of view of the Valence Bond method | the structure|building| of N2 molecule can be represented as follows:
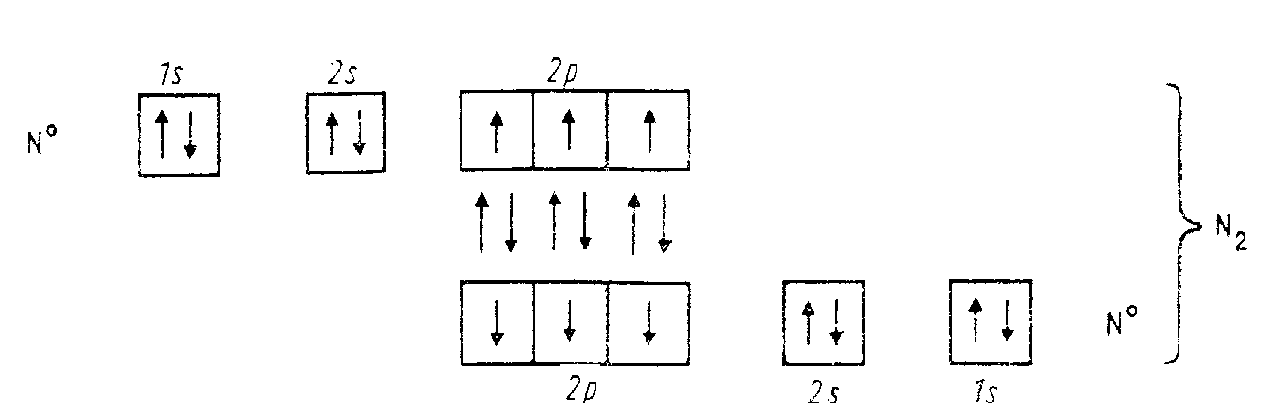
Thus,|on this grow| six unpaired| electrons of 2р-sublevel of both atoms of nitrogen form three electronic dublets| and consequently three identical covalent| bonds|truss|.
The last energy level electronic configuration of this element is 2s22p3. The other elements have vacant d-orbitals and excitement of one of the paired electrons of s-orbital is possible:

 N
P,
As,
Sb, Bi
N
P,
As,
Sb, Bi
This means that maximal covalence of nitrogen (the number of covalent bonds) is four (for instance, there are three -bonds and single delocalised -bond in NO3-ion; the bond order of each N—O bond is less than two and more than one). The oxidation states of nitrogen in compounds are|halving,compound,junction,joint,coupling|: -3, -2, -1, 0 +1, +2, +3, +4, +5.
Phosphorus. The outermost electron shell configuration of the ground state of a phosphorus atom is 3s23p3. As shown above, the availability of vacant d-orbitals changes properties of P significantly although phosphorus is an electronic equivalent of nitrogen (both have identical outermost shells). Phosphorus displays -3, -2 (P2H4), 0, +1, +3, +5 oxidation states. Note that even oxidation states of phosphorus are not typical unlike nitrogen.
Electronegativity (EN) of phosphorus is 2.06. The lower EN, the less typical is the formation of ionic compounds with metals.
Arsenic, Antimony, Bismuth. These elements have the last energy level with 5 electrons - ns2np3, i.e. two paired ns- electrons and three unpaired np-electrons. However, there are also differences in electronic structure between them. Unlike phosphorus, they contain completed (n-1)d-sublevel, and in case of Bi, which is situated after lanthanides, 4f14-sublevel appears.
Forming a chemical bond with three unpaired electrons and depending on the EN of a partner, these elements may display oxidation states -3 and +3. The formation of compounds with the highest oxidation state +5 is attributed to the excited state of the elements. Like other subgroups of p-elements, it becomes less stable with the growth of the period number, i.e. in the series As—Sb—Bi.
Moreover, the secondary periodicity appears in the series As—Sb—Bi due to the presence of inner d- and f-subshells of electrons and their screening effect on outermost shell electrons.
For instance, oxidation state +5 in the middle of the series (Sb) is more stable than in case of As and Bi. The reason of this phenomenon is the increased energy gap between ns- and np-sublevels from As to Bi, and reduced ability to hybridisation of s- and p-orbitals.
Especially big is the difference in case of 6s2- electron pair. Significant penetration of their electron density to the nucleus through the screen of filled completely d-and f-subshells exists there. It is precisely this fact that explains the effect of 6s2 electron pair inertness and therefore, the variety of Bi3 + compounds and very limited number of Bi5+ compounds (they all are very strong oxidants).


 N
N