
- •Course of lectures «Contemporary Physics: Part2»
- •Some Properties of Nuclei
- •Some Properties of Nuclei
- •Some Properties of Nuclei
- •Some Properties of Nuclei
- •Some Properties of Nuclei
- •Some Properties of Nuclei
- •Some Properties of Nuclei
- •Nuclear Binding Energy
- •Nuclear Binding Energy
- •Nuclear Models
- •Nuclear Models
- •Nuclear Models
- •Nuclear Models
- •Radioactivity
- •Radioactivity
- •Radioactivity
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •The Decay Processes
- •Natural Radioactivity
- •Nuclear Reactions
- •Nuclear Reactions
- •Nuclear Magnetic Resonance and
- •Nuclear Magnetic Resonance and
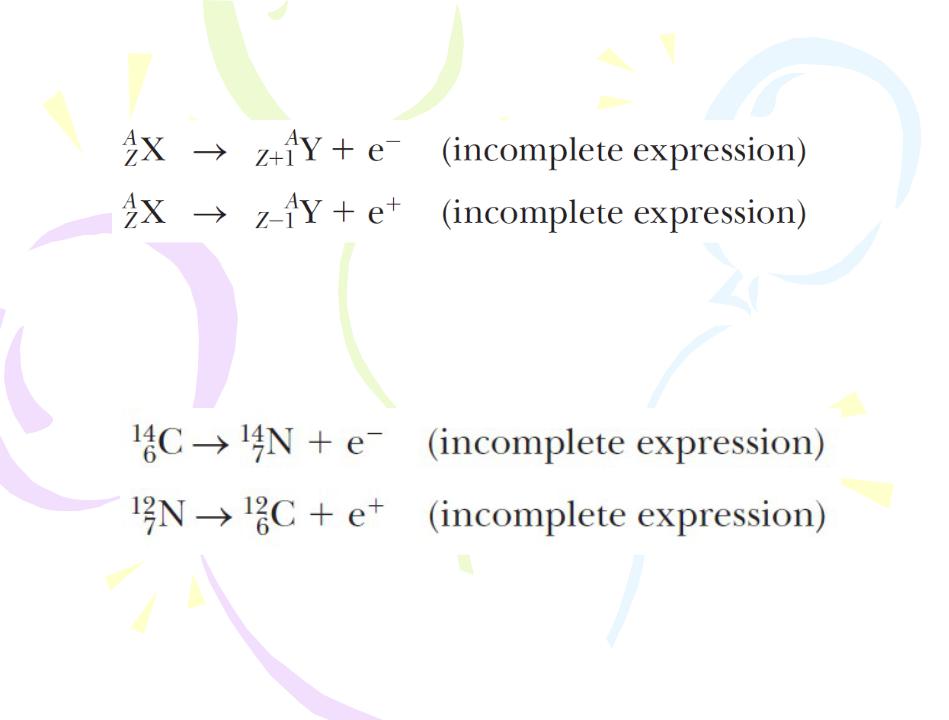
The Decay Processes
Beta Decay
Two typical beta-decay processes are

The Decay Processes
Beta Decay

The Decay Processes
Beta Decay
The neutrino has the following properties:
• It has zero electric charge.
•Its mass is either zero (in which case it travels at the speed of light) or very small; much recent persuasive experimental evidence suggests that the neutrino mass is not zero. Current experiments place the upper bound of the mass of the neutrino at approximately 7 eV/c2.
•It has a spin of 1/2, which allows the law of conservation of angular momentum to be satisfied in beta decay.
•It interacts very weakly with matter and is therefore very difficult to detect.

The Decay Processes
Beta Decay

The Decay Processes
Beta Decay

The Decay Processes
Beta Decay
A process that competes with e+ decay is electron capture:
In most cases, it is a K-shell electron that is captured and the process is therefore referred to as K capture.

The Decay Processes
Gamma Decay

Natural Radioactivity
Radioactive nuclei are generally classified into two groups: (1) unstable nuclei found in nature, which give rise to natural radioactivity, and (2) unstable nuclei produced in the laboratory through nuclear reactions, which exhibit artificial radioactivity.

Nuclear Reactions
The collisions, which change the identity of the target nuclei, are called nuclear reactions. 

the reaction energy Q associated with a nuclear reaction as the difference between the initial and final rest energy resulting from the reaction:
The Q value for this reaction is 17.3 MeV. A reaction such as this one, for which Q is positive, is called exothermic. A reaction for which Q is negative is called endothermic.

Nuclear Reactions
The minimum energy necessary for such a reaction to occur is called the threshold energy. 
If particles a and b in a nuclear reaction are identical so that X and Y are also necessarily identical, the reaction is called a scattering event. If the kinetic energy of the system (a and X) before the event is the same as that of the system (b and Y) after the event, it is classified as elastic scattering. If the kinetic energy of the system after the event is less than that before the event, the reaction is described as inelastic scattering. 
The total number of nucleons before the reaction (1+19=20) is equal to the total number after the reaction (16+4=20). Furthermore, the total charge is the same before (1=9) and after (8=2) the reaction.
