
- •Preface
- •Acknowledgments
- •Contents
- •Contributors
- •1. Introduction
- •2. Evaluation of the Craniomaxillofacial Deformity Patient
- •3. Craniofacial Deformities: Review of Etiologies, Distribution, and Their Classification
- •4. Etiology of Skeletal Malocclusion
- •5. Etiology, Distribution, and Classification of Craniomaxillofacial Deformities: Traumatic Defects
- •6. Etiology, Distribution, and Classification of Craniomaxillofacial Deformities: Review of Nasal Deformities
- •7. Review of Benign Tumors of the Maxillofacial Region and Considerations for Bone Invasion
- •8. Oral Malignancies: Etiology, Distribution, and Basic Treatment Considerations
- •9. Craniomaxillofacial Bone Infections: Etiologies, Distributions, and Associated Defects
- •11. Craniomaxillofacial Bone Healing, Biomechanics, and Rigid Internal Fixation
- •12. Metal for Craniomaxillofacial Internal Fixation Implants and Its Physiological Implications
- •13. Bioresorbable Materials for Bone Fixation: Review of Biological Concepts and Mechanical Aspects
- •14. Advanced Bone Healing Concepts in Craniomaxillofacial Reconstructive and Corrective Bone Surgery
- •15. The ITI Dental Implant System
- •16. Localized Ridge Augmentation Using Guided Bone Regeneration in Deficient Implant Sites
- •17. The ITI Dental Implant System in Maxillofacial Applications
- •18. Maxillary Sinus Grafting and Osseointegration Surgery
- •19. Computerized Tomography and Its Use for Craniomaxillofacial Dental Implantology
- •20B. Atlas of Cases
- •21A. Prosthodontic Considerations in Dental Implant Restoration
- •21B. Overdenture Case Reports
- •22. AO/ASIF Mandibular Hardware
- •23. Aesthetic Considerations in Reconstructive and Corrective Craniomaxillofacial Bone Surgery
- •24. Considerations for Reconstruction of the Head and Neck Oncologic Patient
- •25. Autogenous Bone Grafts in Maxillofacial Reconstruction
- •26. Current Practice and Future Trends in Craniomaxillofacial Reconstructive and Corrective Microvascular Bone Surgery
- •27. Considerations in the Fixation of Bone Grafts for the Reconstruction of Mandibular Continuity Defects
- •28. Indications and Technical Considerations of Different Fibula Grafts
- •29. Soft Tissue Flaps for Coverage of Craniomaxillofacial Osseous Continuity Defects with or Without Bone Graft and Rigid Fixation
- •30. Mandibular Condyle Reconstruction with Free Costochondral Grafting
- •31. Microsurgical Reconstruction of Large Defects of the Maxilla, Midface, and Cranial Base
- •32. Condylar Prosthesis for the Replacement of the Mandibular Condyle
- •33. Problems Related to Mandibular Condylar Prosthesis
- •34. Reconstruction of Defects of the Mandibular Angle
- •35. Mandibular Body Reconstruction
- •36. Marginal Mandibulectomy
- •37. Reconstruction of Extensive Anterior Defects of the Mandible
- •38. Radiation Therapy and Considerations for Internal Fixation Devices
- •39. Management of Posttraumatic Osteomyelitis of the Mandible
- •40. Bilateral Maxillary Defects: THORP Plate Reconstruction with Removable Prosthesis
- •41. AO/ASIF Craniofacial Fixation System Hardware
- •43. Orbital Reconstruction
- •44. Nasal Reconstruction Using Bone Grafts and Rigid Internal Fixation
- •46. Orthognathic Examination
- •47. Considerations in Planning for Bimaxillary Surgery and the Implications of Rigid Internal Fixation
- •48. Reconstruction of Cleft Lip and Palate Osseous Defects and Deformities
- •49. Maxillary Osteotomies and Considerations for Rigid Internal Fixation
- •50. Mandibular Osteotomies and Considerations for Rigid Internal Fixation
- •51. Genioplasty Techniques and Considerations for Rigid Internal Fixation
- •52. Long-Term Stability of Maxillary and Mandibular Osteotomies with Rigid Internal Fixation
- •53. Le Fort II and Le Fort III Osteotomies for Midface Reconstruction and Considerations for Internal Fixation
- •54. Craniofacial Deformities: Introduction and Principles of Management
- •55. The Effects of Plate and Screw Fixation on the Growing Craniofacial Skeleton
- •56. Calvarial Bone Graft Harvesting Techniques: Considerations for Their Use with Rigid Fixation Techniques in the Craniomaxillofacial Region
- •57. Crouzon Syndrome: Basic Dysmorphology and Staging of Reconstruction
- •58. Hemifacial Microsomia
- •59. Orbital Hypertelorism: Surgical Management
- •60. Surgical Correction of the Apert Craniofacial Deformities
- •Index
35
Mandibular Body Reconstruction
Anna-Lisa Söderholm, Dorrit Hallikainen, and Christian Lindqvist
Mandibular continuity defects are the result of surgery for malignant tumors, large benign tumors of various origins (e.g., dental, mandibular bone, surrounding tissues), or different benign diseases (e.g., extensive fibrous dysplasia, cysts or osteomyelitis, and trauma).
A vast majority of these diseases, including cancer,1 involve mainly the mandibular body, the angular area, or both. Mandibular reconstruction surgery is, when appropriate equipment and surgical technique are chosen, usually to be considered as reconstruction of the mandibular body. If the hard tissue framework is successfully restored, good function is achieved; that is in contrast to bridging of even restricted continuity defects in the symphysis area. Lost anterior insertion of muscles of the floor of the mouth and tongue, lost soft tissue equilibrium between the floor of the mouth and lower lip, and lost lip support causes major functional problems for the patient, despite successful contouring of the hard tissue frame. Analogous problems may occur if the condyle is lost. Hence, evaluation of adequate extension of the resection to be performed, without unnecessarily large safety margins, is important for the success of the reconstruction.2
Rehabilitation of patients with mandibular continuity defects has always been a challenging problem. Without adequate primary reconstruction, loss of mandibular continuity, especially if combined with large soft tissue defects, lead to considerable difficulties with speech, mastication, swallowing, and oral continence,3–5 as well as psychosocial problems. Previously, free-bone grafting was the only available method for rebuilding the mandible.3 However, this operation, usually done as a secondary procedure, often led to complica- tions,6–8 and many patients were never offered such a reconstruction.
Today, immediate postoperative rehabilitation of patients with mandibular defects can be achieved through rigid-plate reconstruction. Bone can be transplanted either primarily or in a secondary procedure.9,10
Good early results have been achieved with various plates.11,12 Several problems, including plate exposure, plate
fracture, and infection, however, have occurred during fol- low-up with the various systems used.13,14 However, a majority of these problems can, if detected at an early stage, be solved with good results (Tables 35.1 and 35.2).10,15 Thorough knowledge of the principles and the limits of the system used are essential for good long-term results.15–18
In general, all plate systems work on the same basic principles. A conventional reconstruction plate, developed for osteosynthesis, is fixed to the bone by screws, the heads of which press the plate firmly against the bone surface. The screw holes are oval, and the cross-sectional area of the screw is less than that of the hole. Good buccal bone adaption is needed for adequate fixation. In our experimental studies on sheep, however, resorption of the buccal cortex has been observed to occur under the whole length of large reconstruction plates in almost every case, sometimes restricted to the area under individual screws (Figure 35.1). Adequate fixation was found in histological and microradiographic examination in only 26% of the screws (Table 35.3). A similar resorption was also recorded in our follow-up study on patients resulting in 30% screw failures observed on radiologic examination (Figure 35.2, Table 35.4).15 The plate interferes with blood circulation to the underlying bone, and areas of early osteoporosis are found under rigid plates, corresponding in size to the dimensions of the plate.9,19,20
The conventional plates need four to six screws per fragment if stable fixation is to be guaranteed. Owing to a lack of space, placing this number of screws can be difficult to accomplish in tumor surgery, especially in the ramus-condyle area. Accessory approaches may also be needed, increasing risks of nerve damage and/or scar problems. Loosening of screws and an inadequate number of screws often leads to instability, pain, infection, and a need for plate removal (Figures 35.3 and 35.4).10,14,15
Conventional reconstruction plates, which were originally developed for fracture ostheosynthesis, are in our opinion hazardous for use in permanent mandibular bridging. In reconstructive surgery, the situation is totally different from that in
395

396 |
|
|
|
|
|
A.-L. Söderholm, D. Hallikainen, and C. Lindqvist |
||||
TABLE 35.1 Summary of successfully treated late complications (7 of 34 reconstructions).10 |
|
|
|
|||||||
|
|
|
|
|
|
|
Interval |
|
|
|
|
|
|
|
|
|
|
from |
|
|
Follow-up |
|
|
Diagnosis |
|
Plate used |
|
|
primary |
|
|
from |
|
Age |
(stage of |
Resection |
at primary |
Radiation |
|
surgery |
|
Current |
diagnosis |
Patient |
(years) |
disease) |
performed |
surgery |
(dose) |
Complications |
(month) |
Treatment |
state |
(months) |
|
|
|
|
|
|
|
|
|
|
|
1 |
77 |
SCC (IV) |
Left hemi |
AO-RPC |
Postoperative |
Plate fracture |
24 |
AO-RPC free bone |
FFD |
67 |
|
|
|
|
|
(66 Gy) |
|
|
(iliac crest) |
|
|
2 |
66 |
SCC (IV) |
Right body and |
AO-ARP |
Postoperative |
Screw loosening |
12 |
ARP free bone (iliac |
FFD |
53 |
|
|
|
ramus |
|
(66 Gy) |
proximally |
|
crest) dental |
|
|
|
|
|
|
|
|
|
|
implants |
|
|
3 |
77 |
SCC (IV) |
Left body and |
AO-ARP |
Postoperative |
Screw loosening |
18 |
TH-ARP |
AWD |
44 |
|
|
|
ramus |
|
(66 Gy) |
proximally |
|
|
|
|
4 |
60 |
SCC (IV) |
Symphysis right |
AO-ARP |
Postoperative |
Tumor |
12 |
AO-RPC salvage |
FFD |
41 |
|
|
|
body |
|
(66 Gy) |
recurrence |
|
operation |
|
|
5 |
79 |
SCC (IV) |
Symphysis |
AO-SRP |
Preoperative |
Screw loosening |
22 |
TH-SRP |
FFD |
30* |
|
|
|
|
|
(64 Gy) |
proximally |
|
|
|
|
6 |
26 |
Angiosarcoma |
Symphysis right |
W-RPC |
Postoperative |
Extraoral plate |
3 |
Sternocleido |
DFD |
23† |
|
|
(grade II) |
body and |
|
(66 Gy) |
exposure |
|
musculocutaneous |
|
|
|
|
|
ramus |
|
|
|
|
flap |
|
|
7 |
45 |
SCC (IV) |
Right hemi |
AO-RPC |
Postoperative |
Intraoral plate |
6 |
Antibiotics wound |
FFD |
17† |
|
|
|
|
|
(66 Gy) |
exposure |
|
care |
|
|
Abbreviations: AO-ARP, classic angular AO plate; AO-RPC, classic AO plate with condylar head; AO-SRP, classic straight AO plate; AWD, alive with disease; D, dead from other reason; DFD, dead from disease; FFD, free from disease; SCC, squamous cell carcinoma; TH-ARP, AO-THORP angular plate; TH-SRP, AO-THORP straight plate; W-RPC, Würzburg plate with condylar head.
*From diagnosis of mandibular metastasis, 50-month follow-up from primary diagnosis of SCC of the tongue. †Primary plate in place after soft tissue closure.
Source: From ref. 10.
TABLE 35.2 Summary of patient records for late major complications (7 of 34 reconstructions) resulting in plate removal.10
|
|
|
|
|
|
|
|
|
|
Follow-up |
|
|
Diagnosis |
|
|
|
|
|
Interval to |
|
from |
|
Age |
(stage of |
Resection |
|
Radiation |
|
removal |
State of |
diagnosis |
|
Patient |
(years) |
disease) |
performed |
Plate |
(dose) |
Complication |
(month) |
patient |
(months) |
|
|
|
|
|
|
|
|
|
|
|
|
8 |
48 |
SCC (IV) |
Symphysis |
AO-SRP |
Postoperative |
Infection, fistulation |
12 |
FFD |
77 |
|
|
|
|
|
|
(66 |
Gy) |
|
|
|
|
9 |
60 |
SCC (IV) |
Symphysis |
AO-ARP |
|
— |
Infection, fistulation |
10 |
FFD |
66 |
10 |
57 |
SCC (II) |
Symphysis |
AO-SRP |
|
— |
Fistulation |
17 |
FFD |
34 |
|
|
|
|
|
|
|
Plate exposure (partial |
|
|
|
|
|
|
|
|
|
|
flap necrosis) |
|
|
|
11 |
69 |
SCC (III) |
Right ramus |
AO-ARP |
Postoperative |
Chronic infection, |
6 |
D |
25 |
|
|
|
|
body |
|
(66 |
Gy) |
osteoradionecrosis? |
|
|
|
12 |
77 |
SCC (IV) |
Left hemi |
AO-RCP |
Postoperative |
Chronic infection, |
4 |
FFD |
23 |
|
|
|
|
|
|
(66 |
Gy) |
fistulation |
|
|
|
13 |
78 |
SCC (IV) |
Right ramus |
AO-ARP |
Postoperative |
Tumor recurrence, plate |
7 |
DFD |
16 |
|
|
|
|
body |
|
(66 |
Gy) |
exposure |
|
|
|
14 |
61 |
SCC (IV) |
Right ramus |
AO-ARP |
Preoperative |
Tumor recurrence, |
2 |
DFD |
6 |
|
|
|
|
body |
|
(40 |
Gy) and |
infection |
|
|
|
|
|
|
|
|
postoperative |
|
|
|
|
|
|
|
|
|
|
(30 |
Gy) |
|
|
|
|
Abbreviations: AO-ARP, classic angular AO plate; AO-RPC, classic AO plate with condylar head; AO-SRP, classic straight AO plate; AWD, alive with disease; D, dead from other reason; DFD, dead from disease; FFD, free from disease; SCC, squamous cell carcinoma; TH-ARP, AO-THORP angular plate; TH-SRP, AO-THORP straight plate.
Source: From ref. 10.

35. Mandibular Body Reconstruction |
397 |
a |
b |
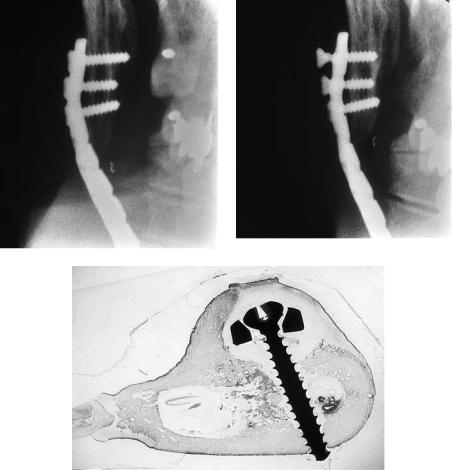
FIGURE 35.1 (a) Resorption under an AO-THORP plate fixed with solid screws for bridging of a continuity defect in a sheep mandible. Detailed image of the plate and the screw fixation in the ramus frag-
fracture treatment, where bone healing at the fracture site enhances stability after 4 to 6 weeks, before buccal resorption extensive enough to jeopardize plate fixation has developed.
The AO-THORP system was developed by Raveh and coworkers.21–23 This reconstruction system allows stable fixation without compression of the plate against the bone surface due to the screw-plate locking principle (Figure 35.5). Good screw adaptation to cortical bone, lingually or buccally, guarantees stability with two or three screws per fragment, as observed in our experimental study on sheep (Figures 35.6 and 35.7)16,17 and radiologic follow-up study of patients (Figure 35.8).15 The fixation stability of this system has proved clearly superior to the conventional plate systems both primarily and in long-term follow-up (Tables 35.4 and 35.5; Figure 35.9). Although permanent reconstruction with bone, dental implants, and bridgework is desirable, plate reconstruction
ment. Slight resorption of the buccal cortex is seen after 8 weeks. Experimental study on sheep (unpublished data). (b) A diagrammatic representation of (a).
can, for several reasons (e.g., patient’s age), be regarded as permanent in many cases. Close monitoring of screw fixation, including adequate radiologic examination, is, however, in such cases required.15
Permanent reconstruction of the mandible requires bony continuity to allow rehabilitation of masticatory function with dental implants and/or dental prostheses. In the case of benign disease, tumor, or traumatic defect, immediate bone grafting is often indicated. For malignant tumors, the timing of the bone transplantation varies. Some surgeons reconstruct the defect with a vascularized bone graft at primary cancer surgery.24 Others prefer a secondary procedure using a strictly extraoral approach after primary wound healing.10,18,25,26 It has also been advocated to wait 1 to 2 years before carrying out the bony reconstruction to ensure early detection of re- currence.26–29 Whichever procedure is preferred, the rigid-
TABLE 35.3 Screw fixation to cortical bone with classic AO screws.
|
|
Number |
Good |
Good |
|
|
Follow-up |
of |
buccal |
lingual |
Adequate |
Sheep |
(weeks) |
screws |
adaptation |
adaptation |
fixation |
|
|
|
|
|
|
A |
5 |
9 |
0 |
0 |
0 |
B |
9 |
9 |
8 |
4 |
8 |
C |
9 |
10 |
0 |
4 |
0 |
D |
14 |
(10) |
(7) |
(8) |
(7)* |
E |
14 |
10 |
2 |
7 |
2 |
Total |
|
38 |
10 |
15 |
10 |
*Plate fracture between the screws excluded from the comparison of screw fixation. Source: From ref. 16.

398 |
A.-L. Söderholm, D. Hallikainen, and C. Lindqvist |
a
b |
|
c |
|
|
|
d
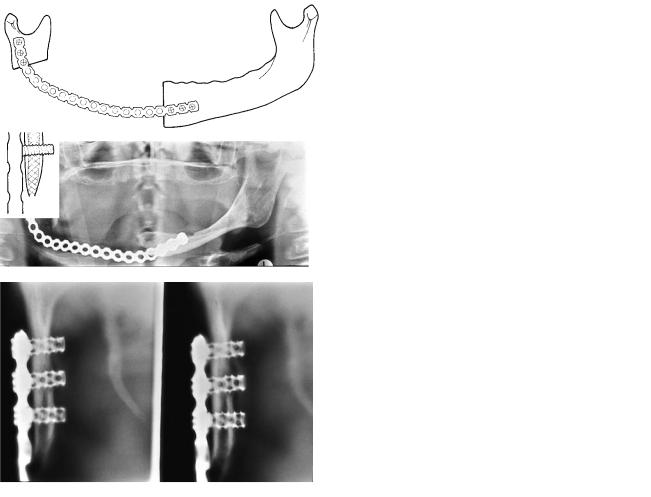
FIGURE 35.2 Reconstruction of the mandibular symphysis area in a 66-year-old woman after cancer ablation surgery. (a) Panoramic view with the plate in place. (b,c) Cross-sectional tomography of the left mandibular body area 5 months postoperatively shows slight re-
sorption of the buccal cortex. (Compare buccal and lingual cortex. Normal cortical thickness is preserved lingually.) (d) A diagrammatic representation of (a), (b), and (c).

35. Mandibular Body Reconstruction |
399 |
TABLE 35.4 Radiologic examination of screw fixation and plate stability in nine cases of mandibular reconstruction using classic AO plates.
|
|
|
|
|
|
|
First signs |
|
|
|
Number of |
Number of |
State of |
Resorption of |
Resorption of |
of screw |
|
|
|
screws |
screws |
screw |
cortical bone |
cortical bone |
loosening |
Follow-up |
Case |
Resection |
proximally |
distally |
fixation |
buccally |
lingually |
(months) |
(months) |
|
|
|
|
|
|
|
|
|
A |
|
3 |
4 |
2 screws |
— |
— |
9 |
55 |
|
|
|
|
loose |
|
|
|
|
B |
|
* |
8 |
good |
xx |
xx |
— |
28 |
C |
|
* |
7 |
1 screw |
— |
— |
28 |
39 |
|
|
|
|
loose |
|
|
|
|
D |
|
* |
5 |
good |
— |
x |
— |
37 |
E |
|
4 |
3 |
3 screws |
xxx |
xxx |
2 |
4 |
|
|
|
|
loose |
|
|
|
|
F |
|
3 |
4 |
3 screws |
xx |
— |
4 |
18 |
|
|
|
|
loose |
|
|
|
|
G |
|
5 |
4 |
4 screws |
— |
— |
20 |
66 |
|
|
|
|
loose |
|
|
|
|
H |
|
3 |
6 |
3 screws |
xxx |
xxx |
5 |
10 |
|
|
|
|
loose |
|
|
|
|
I |
|
4 |
3 |
4 screws |
xxx |
xxx |
8 |
10 |
|
|
|
|
loose |
|
|
|
|
Total |
|
22 |
44 |
20 screw |
5 cases with |
4 cases with |
|
|
|
|
|
|
failures |
resorption |
resorption |
|
|
*Plate with condylar head; x probable resorption; xx slight resorption; xxx moderate resorption. Source: From ref. 14.
plate reconstruction can successfully bridge the defect during bony healing, and if needed, it can be used to fix the graft in place (Figure 35.9).
Vascularized free-bone grafts are being used increasingly to bridge mandibular defects, especially after large resections in cancer surgery. Vascularized grafts are represented by pedicled rib, scapular, or calvarial bone from the vicinity of the mandibular defect, or distant grafts applied using microvascular techniques. Several different donor sites have been used for free vascularized flaps.24,30–37 It seems that iliac crest, fibula, and in some cases the scapula are best suited for mandibular functional reconstruction.35,36,38 However, this technique has limitations with respect to the site and size of the defect, the patient’s general condition, the surgical team available, and the facilities of the clinic. The total failure rate reported for vascularized grafts (11%)35 also has to be taken into account.
Nonvascularized bone grafts are therefore still widely used for mandibular bony reconstruction. Numerous techniques for nonvascularized bone grafting have been presented including
frozen or irradiated autografts from the operation field, alloplastic or bony trays filled with cancellous bone, and compact grafts from the iliac crest.
The introduction of different metal prostheses for primary functional reconstruction has markedly improved the prospects for secondarily performed free-bone grafting.25,28,39 Nevertheless, failure rates of up to 30% have been reported.15,25,29 These failures are, however, often due to the use of an inadequate technique. When nonvascularized bone is used in combination with rigid-plate bridging, the plate has to be fixed in the remaining mandible at both ends. If fixed with one end only in the graft, the plate will loosen during rebuilding of the transplanted bone. The requirement of stability when using this technique cannot be neglected without a high complication risk.
The intraoral approach and intraoral contamination of the operation field have been cited as two reasons for the high failure rates of nonvascularized grafts.25,29,40 Others, however, routinely use the oral approach.27,41 In our own study we found that one third of the cases where an additional

400 |
A.-L. Söderholm, D. Hallikainen, and C. Lindqvist |
a |
b |
c |
d |
e |
f |
FIGURE 35.3 Recurrent carcinoma of the tongue in the left mandibular area, with resection of the mandibular body and reconstruction of the body defect with a classic AO plate. Fixation was performed with three screws proximally. (a) Immediate postoperative panoramic
view. (b) A diagrammatic representation of (a). (c,d) Panoramic detailed images 21 months later shows massive resorption of the angular area. (e,f) A diagrammatic representation of (c) and (d).
35. Mandibular Body Reconstruction |
401 |
a |
b |
c
FIGURE 35.4 (a) Postoperative Townes’ view of proximal (angular) screw fixation of a plate reconstruction in the body region performed at primary cancer surgery. The straight classic plate was fixed at the angle with only three screws. (b) At check-up, 4 years later loosen-
ing of the screws was seen in the x-rays. (c) Histologic view of loose classic AO screw. Buccal cortex resorption under the plate. This screw no longer participates in plate fixation.

402 |
A.-L. Söderholm, D. Hallikainen, and C. Lindqvist |
FIGURE 35.5The screw-plate locking principle ensures adequate plate fixation even with bone apposition only lingually. Histologic view of a section through the head of an AO-THORP hollow screw in position. The expansion bolt presses the head of the screw firmly against the surface of the round plate hole (basic Fuchsin stain, magnification 16).
FIGURE 35.6 Microradiograph of adequately fixed AO-THORP hollow screw (ramus part). No buccal bone apposition left. Good bone apposition to outer screw surface lingually. This is sufficient for adequate screw fixation with this system because of the screw-plate locking principle.
a |
b |
FIGURE 35.7 (a) Microradiograph of adequately fixed AO-THORP hollow screw (ramus part). Good bone apposition both buccally and lingually (magnification 4.4). (b) Histologic view of AO-THORP screw fixed in bone (basic Fuchsin stain; magnification 12.6).
FIGURE 35.8 Comparison of screw fixation failures with classic AO plates and AO-THORP reconstruction in long-term follow-up.15

35. Mandibular Body Reconstruction |
|
|
|
|
|
403 |
||
TABLE 35.5 Radiological examination of screw fixation and plate stability in 13 cases of mandibular reconstructions using the |
|
|||||||
AO-THORP system.15 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
First signs |
|
|
|
Number of |
Number of |
State of |
Resorption of |
Resorption of |
of screw |
|
|
|
screws |
screws |
screw |
cortical bone |
cortical bone |
loosening |
Follow-up |
Case |
Resection |
(ramus) |
(body) |
fixation |
buccally |
lingually |
(months) |
(months) |
|
|
|
|
|
|
|
|
|
K |
|
3 |
3 |
good |
— |
— |
— |
6 |
L |
|
2 |
2 |
2 screw |
x* |
— |
1 |
4 |
|
|
|
|
fractures |
|
|
|
|
H |
|
3 |
5 |
good |
xx |
— |
— |
16 |
M |
|
3 |
3 |
good |
xx |
xx |
— |
33 |
N |
|
3 |
3 |
good |
xx |
— |
— |
5 |
O |
|
4 |
4 |
good |
xx |
— |
— |
13 |
P |
|
2 |
4 |
good |
xx |
— |
— |
12 |
Q |
|
2 |
3 |
good |
— |
— |
— |
9 |
R |
|
3 |
3 |
good |
x |
x |
— |
31 |
S |
† |
3 |
3 |
1 screw |
xx† |
— |
3 |
9 |
|
|
|
|
loosened |
|
|
|
|
T |
|
2 |
3 |
good |
xx |
— |
— |
9 |
F |
|
3 |
3 |
good |
— |
— |
— |
35 |
V |
|
2 |
2 |
good |
x |
— |
— |
12 |
Total |
|
35 |
41 |
3 screw |
10 cases with |
2 cases with |
|
|
|
|
|
|
failures |
resorption |
resorption |
|
|
*Tumor recurrence at fixation area.
† secondary reconstruction after radiotherapy. x probable resorption; xx slight resorption.
404 |
A.-L. Söderholm, D. Hallikainen, and C. Lindqvist |
a |
b |
c |
d |
e |
f |
FIGURE 35.9 A segmental resection of the right angle and body was performed due to mandibular cancer in a 55-year-old woman. After postoperative radiotherapy the patient was referred 1 year later to our department for rehabilitation. (a) Panoramic view at referral. (b) A diagrammatic representation of (a). (c) Reconstruction of the
mandibular continuity was performed with an angular AO-THORP plate. (d) A diagrammatic representation of (c). (e) Cross-sectional tomography of the condylar neck area 36 months later. The screws are in good position, and no bone resorption has occurred. (f) A diagrammatic representation of (e).

35. Mandibular Body Reconstruction |
405 |
g |
h |
i |
j |
k |
l |
FIGURE 35.9 Continued. (g) In a second operation, nonvascular bone from the iliac crest was transplanted to the defect area. Soft tissue shortage was reconstructed by a pectoralis major flap. (h) A diagrammatic representation of (g). (i) Dental implants were thereafter installed in the symphysis area, both in the bone transplant to the right and in the patient’s own mandibular bone to the left. The plate
was removed from the body area. (j) Intraoral view 4 years later with the dental implants and the connecting bar. (k) The patient’s implant based over dentures in good occlusion and (l) mouth opening of the patient. Both speech and masticatory function have improved markedly in consequence of the reconstruction performed.

406 |
A.-L. Söderholm, D. Hallikainen, and C. Lindqvist |
a |
b |
c |
d |
e |
f |
FIGURE 35.10 Because of an ameloblastoma in the right angular area, a segmental resection of the mandible was necessary. Primary reconstruction with an AO-THORP plate and nonvascular bone from the iliac crest was performed. (a) Immediate preoperative panoramic view. (b) A diagrammatic representation of (a). (c) Postoperative de-
tailed image of the plate and the one-piece bone reconstruction.
(d) A diagrammatic representation of (c). (e) Due to intraoral wound dehiscence, infection of the resection area and severe resorption of the bone transplant followed. Finally, the transplant was lost. (f) A diagrammatic representation of (e).

35. Mandibular Body Reconstruction |
407 |
g |
h |
i |
j |
FIGURE 35.10 Continued. (g) After revision, antibiotics, and secondary healing, a new similar nonvascular transplant was inserted several months later from a strictly extraoral approach. (h) A diagrammatic representation of (g). (i) The height of the bone trans-
intraoral approach was needed resulted in postoperative infection and graft failure.15 In our opinion, these complications stress the importance of extremely good intraoral wound care and the use of an extraoral approach only whenever possible (Figure 35.10).
High plate rigidity has been claimed to enhance graft resorption. Plate removal within 6 months after bone grafting has been recommended.25,42 This was not confirmed in our own study. The mean interval between bone grafting and plate removal was 26 months for the patients with good bone healing and a resorption of less than 15%.15
Nonvascularized bone grafts in combination with rigidplate reconstruction still play an important role in permanent mandibular reconstruction (Figure 35.11).15,43 The technique does not require much time or resources and is particularly valuable in trauma and benign tumor cases.
Regular radiologic examination is essential in graft fol- low-up. Radiologic examination contributes to the evaluation of the possible resorption and resorption rate. Complications can be detected early and rescue therapy initiated wtih adequate time. Special attention must be paid to the imaging technique and image quality (Figure 35.2b,c). Good spatial resolution and good contrast are necessary, and the
plant was unchanged 3 years later. The plate has been removed. Three of the hollow screws were integrated into the bone tissue and could not be removed by a screwdriver and hence were left. (j) A diagrammatic representation of (i).
imaging procedure must be selected according to the specified diagnostic task. Radiologic literature on bone graft evaluation is sparse; especially, experience with computed tomography (CT) and magnetic resonance imaging (MRI) is limited.44,45 Artifacts originating from metal reconstruction devices may complicate interpretation of CT images. MRI is even more sensitive to minute magnetic metal pieces, such as those originating from drills or other metal tools. We have used panoramic radiography in combination with detailed panoramic images and cross-sectional tomography.14,18 Close collaboration between clinician and radiologist is beneficial.
Editors Note
Concerning two-screw fixation of segments: Following J. Raveh’s initial preference for the fixation of segments with only 2 hollow screws (owing to the possibilities for bone ingrowth), international clinical experience presently indicates the necessity for a minimum of 3 to 4 screws (hollow or otherwise). The exception remains that 2-screw fixation of only the subcondylar segment is acceptable. For further discussion see Chapter 27.

a |
b |
c |
d |
e |
f |
g
FIGURE 35.11 A large ameloblastoma of the right mandibular angle in a 37-year-old woman was treated by a segmental resection and reconstruction with an AO classic angular plate and multiple nonvascular bone transplants from the iliac crest. (a) A panoramic view of the reconstruction. (b) A diagrammatic representation of
(a). (c) The patient’s mandible after removal of the plate. (d) A diagrammatic representation of (c). (e,f) Rehabilitation of the dentition was performed by dentures. (g) Excellent occlusion, mouth opening, swallowing, mastication, and speech were achieved.
35. Mandibular Body Reconstruction
References
1.Söderholm A-L. Oral carcinoma of the mandibular region. Br J Oral Maxillofac Surg. 1990;28:383–389.
2.Söderholm A-L, Lindqvist C, Hietanen J, et al. Bone scanning for evaluating mandibular bone extension of oral squamous cell carcinoma. J Oral Maxillofac Surg. 1990;48:252–257.
3.Cohen M, Schoultz RC. Mandibular reconstruction. Symposium on head and neck surgery. Clin Plast Surg. 1985;12:411–422.
4.McQuarrie DG. Oral cancer. In: McQuarrie DG, Adams GL, Shons AR, Brown GA, eds. Head and Neck Cancer. Clinical Decisions and Management Principles. Chicago: Year Book Medical Publishers, Inc.; 1986.
5.Westin T, Jansson A, Zenckert C, et al. Mental depression is associated with malnutrition in patients with head and neck cancer. Arch Otolaryngol Head Neck Surg. 1988;114:1449–1453.
6.Höltje W-J, Lendtrodt J. Infektionen autologer Knochentransplantate nach Defektrekonstruktion des Unterkiefers. Fortschr Kiefer Gesichtschir. 1976;20:32–35.
7.Sailer HF. Ergebnisse der gleichzeitigen Resektion und Rekonstruktion des Unterkiefers auf oralem Weg. Fortschr Kiefer Gesichtschir. 1976;20:45–46.
8.Duncan MJ, Manktelow RT, Zuker, et al. Mandibular reconstruction in the radiated patient: the role of osteocutaneous free tissue transfers. Plast Reconstr Surg. 1985;76:829–840.
9.Spiessl B. Internal Fixation of the Mandible. A Manual of AO/ASIF Principles. New York: Springer-Verlag; 1989.
10.Lindqvist C, Söderholm A-L, Laine P, et al. Rigid reconstruction plates for immediate reconstruction following mandibular resection for malignant tumors. J Oral Maxillofac Surg. 1992;50:1158–1163.
11.Schmoker RR. Mandibular reconstruction using a special plate. Animal experiments and clinical application. J Maxillofac Surg. 1983;11:99–106.
12.Söderholm A-L, Lindqvist C, Laine P, et al. Primary reconstruction of the mandible in cancer surgery. A report of 13 reconstructions according to the principles of rigid internal fixation. Int J Oral Maxillofac Surg. 1988;17:194–197.
13.Platz H, Falkensammer G, Hudec M. Zur Problematik der Defektüberbrückung nach Unterkieferresektionen mit Metallimplantaten. Dtsch Z Mund Kiefer Gesichtschir. 1987;11:269–275.
14.Komisar A, Warman S, Danzinger E. A critical analysis of immediate and delayed mandibular reconstruction using AO plates.
Arch Otolaryngol Head Neck Surg. 1989;115:830–833.
15.Söderholm A-L, Hallikainen D, Lindqvist C. Long-term stability of two different mandibular bridging systems. Arch Otolaryngol Head Neck Surg. 1993;119:1031–1036.
16.Söderholm, A-L, Lindqvist C, Skutnabb K, et al. Bridging of mandibular defects with two different reconstruction systems: An experimental study. J Oral Maxillofac Surg. 1991;49:1098– 1105.
17.Söderholm A-L, Rahn BA, Suutnabb K, Lindqvist C. Fixation with reconstruction plates under critical conditions: The role of screw characteristics. Int J Oral Maxillofac Surg. 1996;25: 469–473.
18.Söderholm A-L, Hallikainen D, Lindqvist C. Radiological fol- low-up of bone transplants for bridging mandibular continuity defects. Oral Surg Med Pathol. 1992;73:253–261.
19.Matter P, Brennwald J, Perren SM. Biologische reaktion des
409
Knochens aus Osteosyntheseplatte. Helv Chir Acta. 1974;1:1–40.
20.Gunst MA. Interference with bone blood supply through plating of intact bone. In: Uthoff HK, ed. Current Concepts of Internal Fixation of Fractures. New York: Springer-Verlag; 1980: 268–276.
21.Raveh J, Sutter F, Hellem S. Surgical procedures for reconstruction of the lower jaw using the titanium-coated hollowscrew reconstruction plate system: bridging of defects. Otolaryngol Clin North Am. 1987;20:535–558.
22.Raveh J. Lower jaw reconstruction with the THORP system for bridging of lower jaw defects. Head Neck Cancer. 1990;II:344– 349.
23.Sutter F, Raveh J. Titanium-coated hollow screw and reconstruction plate system for bridging of lower jaw defects: biomechanical aspects. Int J Oral Maxillofac Surg. 1988;17:267– 274.
24.Wenig BL, Keller AJ. Rigid internal fixation and vascularized bone grafting in mandibular reconstruction. Clin Plast Surg. 1989;16:125–131.
25.Krüger E, Krumholz K. Results of bone grafting after rigid fixation. J Oral Maxillofac Surg. 1984;42:491–496.
26.Esser E, Montag H. Konventionelle Transplantatchirurgie und enosseale Impantate: Ein Behandlungskonzept zur Rehabilation nach radikaler Tumorchirurgie der unteren Mundhölenetage.
Dtsch Z Mund Kiefer Gesichtschir. 1987;11:77–87.
27.Stoll P, Schilli W. Long-term follow up of donor and recipient sites after autologous bone grafts for reconstruction of the facial skeleton. J Oral Maxillofac Surg. 1981;39:676–677.
28.Egyedi P. Wound infection after mandibular reconstruction with autogenous graft. Ann Acad Med Singapore. 1986;15:340–345.
29.Krumholz K. Rekonstruktion des Unterkiefers und Oberkiefers mit Knochen: Ein Bericht über 115 Fälle. Dtsch Z Mund Kiefer Gesichtschir. 1987;11:408–416.
30.Taylor GI, Townsend P, Corlett R. Superiority of the deep circumflex iliac vessels as the supply for free groin flaps: experimental work. Plast Reconstr Surg. 1979;64:595–604.
31.Baker SR. Reconstruction of mandibular defects with the vascularized free tensor fascia lata osteomyocutaneous flap. Arch Otolaryngol. 1981;107:414–418.
32.Rosen IB, Mankeltow RT, Zuker RM, et al. Application of microvascular free osteocutaneous flaps in the management of postradiation recurrent oral cancer. Am J Surg. 1985;150:474–479.
33.Soutar DS, McGregor IA. The radial forearm in intraoral reconstruction: the experience of 60 consecutive cases. Plast Reconstr Surg. 1986;78:1–8.
34.Zuker RM, Mankeltow RT. The dorsalis pedis free flap: technique of elevation, foot closure, and flap application. Plast Reconstr Surg. 1986;77:93–102.
35.David DJ, Tan E, Katsaros J, et al. Mandibular reconstruction with vascularized iliac crest: a 10-year experience. Plast Reconstr Surg. 1988;82:792–801.
36.Lyberg T, Olstad OA. The vascularized fibular flap for mandibular reconstruction. J Craniomaxillofac Surg. 1991;19:113–118.
37.Urken ML. Composite free flaps in oromandibular reconstruction. Review of the literature. Arch Otolaryngol Head Neck Surg. 1991;117:724–732.
38.Fig LM, Shulkin BL, Sullivan MJ, et al. Utility of emission tomography in evaluation of mandibular bone grafts. Arch Otolaryngol Head Neck Surg. 1990;116:191–196.
410
39.Spiessl B. A new method of anatomical reconstruction of extensive defects of the mandible with autogenous cancellous bone. J Craniomaxillofac Surg. 1980;8:78–83.
40.Lawson W, Biller HF. Mandibular reconstruction: bone graft techniques. Otolaryngol Head Neck Surg. 1982;90:589– 594.
41.Obwegeser HL, Häussler F, Ibarra E. Behandlung des infizierten Knochentransplantates bei der Unterkieferrekonstruktion.
Fortschr Kiefer Gesichtschir. 1984;29:76–77.
A.-L. Söderholm, D. Hallikainen, and C. Lindqvist
42.Lentrodt J, Fritzmeier CU, Bethmann I. Beitrag zur osteoplastischen Rekonstruktion des Unterkiefers. Dtsch Z Mund Kiefer Gesichtschir. 1985;9:5–19.
43.Piggot TA, Logan AM. Mandibular reconstruction by “simple” bone graft. Br J Plast Surg. 1983;36:9–15.
44.Bowerman JW, Huges LJ. Radiology of bone grafts. Radiol Clin North Am. 1975;13:67–77.
45.Kattapuram SV, Phillips WC, Mankin HJ. Intercalary bone allografts; radiographic evaluation. Radiology. 1989;179:137–141.
