
- •Contents
- •Preface to the first edition
- •Flagella
- •Cell walls and mucilages
- •Plastids
- •Mitochondria and peroxisomes
- •Division of chloroplasts and mitochondria
- •Storage products
- •Contractile vacuoles
- •Nutrition
- •Gene sequencing and algal systematics
- •Classification
- •Algae and the fossil record
- •REFERENCES
- •CYANOPHYCEAE
- •Morphology
- •Cell wall and gliding
- •Pili and twitching
- •Sheaths
- •Protoplasmic structure
- •Gas vacuoles
- •Pigments and photosynthesis
- •Akinetes
- •Heterocysts
- •Nitrogen fixation
- •Asexual reproduction
- •Growth and metabolism
- •Lack of feedback control of enzyme biosynthesis
- •Symbiosis
- •Extracellular associations
- •Ecology of cyanobacteria
- •Freshwater environment
- •Terrestrial environment
- •Adaption to silting and salinity
- •Cyanotoxins
- •Cyanobacteria and the quality of drinking water
- •Utilization of cyanobacteria as food
- •Cyanophages
- •Secretion of antibiotics and siderophores
- •Calcium carbonate deposition and fossil record
- •Chroococcales
- •Classification
- •Oscillatoriales
- •Nostocales
- •REFERENCES
- •REFERENCES
- •REFERENCES
- •RHODOPHYCEAE
- •Cell structure
- •Cell walls
- •Chloroplasts and storage products
- •Pit connections
- •Calcification
- •Secretory cells
- •Iridescence
- •Epiphytes and parasites
- •Defense mechanisms of the red algae
- •Commercial utilization of red algal mucilages
- •Reproductive structures
- •Carpogonium
- •Spermatium
- •Fertilization
- •Meiosporangia and meiospores
- •Asexual spores
- •Spore motility
- •Classification
- •Cyanidiales
- •Porphyridiales
- •Bangiales
- •Acrochaetiales
- •Batrachospermales
- •Nemaliales
- •Corallinales
- •Gelidiales
- •Gracilariales
- •Ceramiales
- •REFERENCES
- •Cell structure
- •Phototaxis and eyespots
- •Asexual reproduction
- •Sexual reproduction
- •Classification
- •Position of flagella in cells
- •Flagellar roots
- •Multilayered structure
- •Occurrence of scales or a wall on the motile cells
- •Cell division
- •Superoxide dismutase
- •Prasinophyceae
- •Charophyceae
- •Classification
- •Klebsormidiales
- •Zygnematales
- •Coleochaetales
- •Charales
- •Ulvophyceae
- •Classification
- •Ulotrichales
- •Ulvales
- •Cladophorales
- •Dasycladales
- •Caulerpales
- •Siphonocladales
- •Chlorophyceae
- •Classification
- •Volvocales
- •Tetrasporales
- •Prasiolales
- •Chlorellales
- •Trebouxiales
- •Sphaeropleales
- •Chlorosarcinales
- •Chaetophorales
- •Oedogoniales
- •REFERENCES
- •REFERENCES
- •EUGLENOPHYCEAE
- •Nucleus and nuclear division
- •Eyespot, paraflagellar swelling, and phototaxis
- •Muciferous bodies and extracellular structures
- •Chloroplasts and storage products
- •Nutrition
- •Classification
- •Heteronematales
- •Eutreptiales
- •Euglenales
- •REFERENCES
- •DINOPHYCEAE
- •Cell structure
- •Theca
- •Scales
- •Flagella
- •Pusule
- •Chloroplasts and pigments
- •Phototaxis and eyespots
- •Nucleus
- •Projectiles
- •Accumulation body
- •Resting spores or cysts or hypnospores and fossil Dinophyceae
- •Toxins
- •Dinoflagellates and oil and coal deposits
- •Bioluminescence
- •Rhythms
- •Heterotrophic dinoflagellates
- •Direct engulfment of prey
- •Peduncle feeding
- •Symbiotic dinoflagellates
- •Classification
- •Prorocentrales
- •Dinophysiales
- •Peridiniales
- •Gymnodiniales
- •REFERENCES
- •REFERENCES
- •Chlorarachniophyta
- •REFERENCES
- •CRYPTOPHYCEAE
- •Cell structure
- •Ecology
- •Symbiotic associations
- •Classification
- •Goniomonadales
- •Cryptomonadales
- •Chroomonadales
- •REFERENCES
- •CHRYSOPHYCEAE
- •Cell structure
- •Flagella and eyespot
- •Internal organelles
- •Extracellular deposits
- •Statospores
- •Nutrition
- •Ecology
- •Classification
- •Chromulinales
- •Parmales
- •Chrysomeridales
- •REFERENCES
- •SYNUROPHYCEAE
- •Classification
- •REFERENCES
- •EUSTIGMATOPHYCEAE
- •REFERENCES
- •PINGUIOPHYCEAE
- •REFERENCES
- •DICTYOCHOPHYCEAE
- •Classification
- •Rhizochromulinales
- •Pedinellales
- •Dictyocales
- •REFERENCES
- •PELAGOPHYCEAE
- •REFERENCES
- •BOLIDOPHYCEAE
- •REFERENCE
- •BACILLARIOPHYCEAE
- •Cell structure
- •Cell wall
- •Cell division and the formation of the new wall
- •Extracellular mucilage, biolfouling, and gliding
- •Motility
- •Plastids and storage products
- •Resting spores and resting cells
- •Auxospores
- •Rhythmic phenomena
- •Physiology
- •Chemical defense against predation
- •Ecology
- •Marine environment
- •Freshwater environment
- •Fossil diatoms
- •Classification
- •Biddulphiales
- •Bacillariales
- •REFERENCES
- •RAPHIDOPHYCEAE
- •REFERENCES
- •XANTHOPHYCEAE
- •Cell structure
- •Cell wall
- •Chloroplasts and food reserves
- •Asexual reproduction
- •Sexual reproduction
- •Mischococcales
- •Tribonematales
- •Botrydiales
- •Vaucheriales
- •REFERENCES
- •PHAEOTHAMNIOPHYCEAE
- •REFERENCES
- •PHAEOPHYCEAE
- •Cell structure
- •Cell walls
- •Flagella and eyespot
- •Chloroplasts and photosynthesis
- •Phlorotannins and physodes
- •Life history
- •Classification
- •Dictyotales
- •Sphacelariales
- •Cutleriales
- •Desmarestiales
- •Ectocarpales
- •Laminariales
- •Fucales
- •REFERENCES
- •PRYMNESIOPHYCEAE
- •Cell structure
- •Flagella
- •Haptonema
- •Chloroplasts
- •Other cytoplasmic structures
- •Scales and coccoliths
- •Toxins
- •Classification
- •Prymnesiales
- •Pavlovales
- •REFERENCES
- •Toxic algae
- •Toxic algae and the end-Permian extinction
- •Cooling of the Earth, cloud condensation nuclei, and DMSP
- •Chemical defense mechanisms of algae
- •The Antarctic and Southern Ocean
- •The grand experiment
- •Antarctic lakes as a model for life on the planet Mars or Jupiter’s moon Europa
- •Ultraviolet radiation, the ozone hole, and sunscreens produced by algae
- •Hydrogen fuel cells and hydrogen gas production by algae
- •REFERENCES
- •Glossary
- •Index
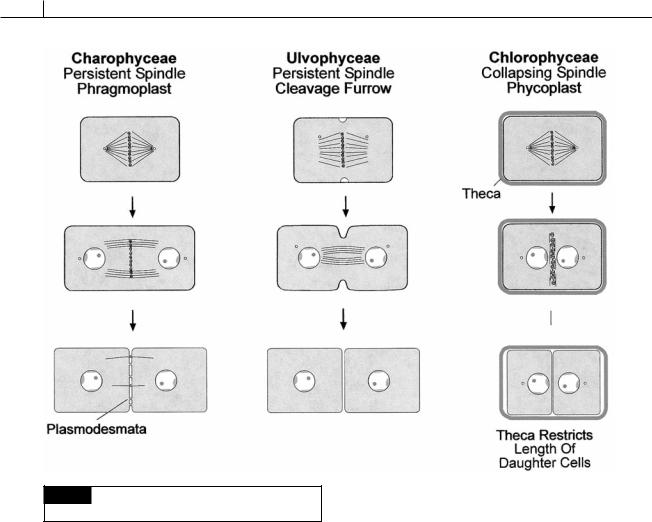
148 EVOLUTION OF THE CHLOROPLAST
Fig. 5.8 Schematic drawing of the type of cell division in
the Charophyceae, Ulvophyceae, and Chlorophyceae.
Cell division
Two types of interzonal spindles occur in telophase cells of the Chlorophyta: the persistent type and the collapsing type. The new cell wall can be formed between the daughter cells by means of a phragmoplast, a cleavage furrow, or a phycoplast. Three of the classes in the Chlorophyta are characterized by the type of spindle in the class and the way the new cross wall is formed (Fig. 5.8).
Persistent interzonal spindle and phragmoplast (Charophyceae)
In the Charophyceae, the microtubular spindle persists even after the daughter nuclei have separated in telophase. The daughter nuclei are separated by the length of the persistent spindle while a new cross wall is formed by a phragmoplast in the more advanced members. Wall formation by a
phragmoplast initially involves the production of vesicles by the dictyosomes. The vesicles contain the components of the new cross walls. The persistent spindle microtubules may function in guiding the vesicles to the area of the new cross wall which will separate the two daughter cells. The vesicles fuse, releasing their contents which form the new cross wall. Plasmodesmata are formed in the cross wall between the daughter cells where the persistent spindle microtubules traverse the cross wall. In some of the primitive Charophyceae, the new cross wall is formed by a phragmoplast in association with an infurrowing of the plasma membrane. In the more advanced Charophyceae (those in the Coleochaetales and Charales), the cross wall is formed only by a phragmoplast.
Persistent interzonal spindle and a cleavage furrow (Ulvophyceae)
In the Ulvophyceae, the interzonal spindle persists during telophase, holding the daughter

CHLOROPHYTA 149
cells apart while the new cross wall is formed by an infurrowing of the plasma membrane. As the plasma membrane furrows inward, dictyosome vesicles fuse with the plasma membrane, behind the infurrowing, producing the new cross wall.
Collapsing interzonal spindle and a cleavage furrow (Chlorophyceae)
In the Chlorophyceae, the mitotic spindle collapses after nuclear division. This results in the two daughter nuclei coming close together in telophase because there is no longer a persistent interzonal spindle to hold the nuclei apart. The position of the new cross walls becomes outlined by microtubules of the phycoplast that arise perpendicular to the former position of the spindle microtubules. Dictyosome vesicles fuse between the phycoplast microtubules, forming the new cross wall. The Chlorophyceae have motile cells with a cell wall (theca) and thus differ from the Ulvophyceae and Charophyceae. The phycoplast evolved in conjunction with the production of cell walls in these algae (Mattox and Stewart, 1977). During cell division in cells with a persistent spindle (in the Prasinophyceae, Charophyceae, and Ulvophyceae), there is extensive elongation of the cell during anaphase. This cell elongation presents no problem in naked cells or cells covered with scales. However, in the Chlorophyceae the cells are not easily able to elongate in response to the anaphase elongation of the persistent spindle. The reason is that the Chlorophyceae have walls around the motile cells. Therefore, evolution of the phycoplast and collapsing spindle, which does not involve rapid elongation of the daughter cells, presents an evolutionary advantage.
Glycolate degradation
Glycolate, the major substrate of photorespiration, is derived from phosphoglycolate, which is formed by the oxygenation of ribulose-1,5-diphosphate:
ribulose-1,5- |
→ |
COO |
→ |
COO |
||
diphosphate |
|
|
|
|
||
|
phosphoglycerate |
CH |
OPO2 |
CH |
OH |
|
|
2 |
2 |
|
3 |
2 |
|
|
|
phosphoglycolate |
glycolate |
|||
The glycolate is metabolized |
in microbodies |
|||||
called peroxisomes. In the peroxisomes, glycolate
is oxidized to glyoxylate. The H2O2 produced in the reaction is cleaved by the enzyme catalase to H2O and O2:
COO |
O2 |
H2O2 |
COO |
|
|
||
|
|
||
|
|
||
CH2OH → C H |
|||
glycolate |
|
|
|
O glyoxylate
The above oxidation of glycolate can be catalyzed by the enzyme glycolate dehydrogenase or the enzyme glycolate oxidase (Gruber et al., 1974). In the Charophyceae, the reaction is catalyzed by glycolate oxidase, whereas in the Chlorophyceae and the Ulvophyceae, the reaction is catalyzed by glycolate dehydrogenase (Suzuki et al., 1991). Glycolate oxidase probably represents the primitive condition since Cyanophora paradoxa in the Glaucophyta catalyzes the reaction with this enzyme (Betsche et al., 1992).
Urea degradation
In the Charophyceae, Ulvophyceae, and higher plants, urea is broken down by the enzyme urease:
|
O |
|
|
|
|
|
|
|
|
H |
N C NH |
2H O → 2NH CO |
2 |
|
2 |
2 |
2 |
4 |
|
|
urea |
|
urease |
|
In the Chlorophyceae, urea is broken down by the enzyme urea amidolyase (Syrett and Al-Houty, 1984):
O
|
|
Mg2 , K |
|
H |
N C NH |
ATP 2NH HCO |
|
2 |
2 |
4 |
3 |
|
urea |
HCO |
|
3 |
ADP Pi |
urea |
|
amidolyase |
|
Superoxide dismutase
Superoxide dismutases (SOD) are a group of enzymes that catalyze the reaction:
O2 O2 2H → H2O2 O2
Superoxide dismutases are important because they take highly reactive, potentially damaging
