
- •Contents
- •Preface to the first edition
- •Flagella
- •Cell walls and mucilages
- •Plastids
- •Mitochondria and peroxisomes
- •Division of chloroplasts and mitochondria
- •Storage products
- •Contractile vacuoles
- •Nutrition
- •Gene sequencing and algal systematics
- •Classification
- •Algae and the fossil record
- •REFERENCES
- •CYANOPHYCEAE
- •Morphology
- •Cell wall and gliding
- •Pili and twitching
- •Sheaths
- •Protoplasmic structure
- •Gas vacuoles
- •Pigments and photosynthesis
- •Akinetes
- •Heterocysts
- •Nitrogen fixation
- •Asexual reproduction
- •Growth and metabolism
- •Lack of feedback control of enzyme biosynthesis
- •Symbiosis
- •Extracellular associations
- •Ecology of cyanobacteria
- •Freshwater environment
- •Terrestrial environment
- •Adaption to silting and salinity
- •Cyanotoxins
- •Cyanobacteria and the quality of drinking water
- •Utilization of cyanobacteria as food
- •Cyanophages
- •Secretion of antibiotics and siderophores
- •Calcium carbonate deposition and fossil record
- •Chroococcales
- •Classification
- •Oscillatoriales
- •Nostocales
- •REFERENCES
- •REFERENCES
- •REFERENCES
- •RHODOPHYCEAE
- •Cell structure
- •Cell walls
- •Chloroplasts and storage products
- •Pit connections
- •Calcification
- •Secretory cells
- •Iridescence
- •Epiphytes and parasites
- •Defense mechanisms of the red algae
- •Commercial utilization of red algal mucilages
- •Reproductive structures
- •Carpogonium
- •Spermatium
- •Fertilization
- •Meiosporangia and meiospores
- •Asexual spores
- •Spore motility
- •Classification
- •Cyanidiales
- •Porphyridiales
- •Bangiales
- •Acrochaetiales
- •Batrachospermales
- •Nemaliales
- •Corallinales
- •Gelidiales
- •Gracilariales
- •Ceramiales
- •REFERENCES
- •Cell structure
- •Phototaxis and eyespots
- •Asexual reproduction
- •Sexual reproduction
- •Classification
- •Position of flagella in cells
- •Flagellar roots
- •Multilayered structure
- •Occurrence of scales or a wall on the motile cells
- •Cell division
- •Superoxide dismutase
- •Prasinophyceae
- •Charophyceae
- •Classification
- •Klebsormidiales
- •Zygnematales
- •Coleochaetales
- •Charales
- •Ulvophyceae
- •Classification
- •Ulotrichales
- •Ulvales
- •Cladophorales
- •Dasycladales
- •Caulerpales
- •Siphonocladales
- •Chlorophyceae
- •Classification
- •Volvocales
- •Tetrasporales
- •Prasiolales
- •Chlorellales
- •Trebouxiales
- •Sphaeropleales
- •Chlorosarcinales
- •Chaetophorales
- •Oedogoniales
- •REFERENCES
- •REFERENCES
- •EUGLENOPHYCEAE
- •Nucleus and nuclear division
- •Eyespot, paraflagellar swelling, and phototaxis
- •Muciferous bodies and extracellular structures
- •Chloroplasts and storage products
- •Nutrition
- •Classification
- •Heteronematales
- •Eutreptiales
- •Euglenales
- •REFERENCES
- •DINOPHYCEAE
- •Cell structure
- •Theca
- •Scales
- •Flagella
- •Pusule
- •Chloroplasts and pigments
- •Phototaxis and eyespots
- •Nucleus
- •Projectiles
- •Accumulation body
- •Resting spores or cysts or hypnospores and fossil Dinophyceae
- •Toxins
- •Dinoflagellates and oil and coal deposits
- •Bioluminescence
- •Rhythms
- •Heterotrophic dinoflagellates
- •Direct engulfment of prey
- •Peduncle feeding
- •Symbiotic dinoflagellates
- •Classification
- •Prorocentrales
- •Dinophysiales
- •Peridiniales
- •Gymnodiniales
- •REFERENCES
- •REFERENCES
- •Chlorarachniophyta
- •REFERENCES
- •CRYPTOPHYCEAE
- •Cell structure
- •Ecology
- •Symbiotic associations
- •Classification
- •Goniomonadales
- •Cryptomonadales
- •Chroomonadales
- •REFERENCES
- •CHRYSOPHYCEAE
- •Cell structure
- •Flagella and eyespot
- •Internal organelles
- •Extracellular deposits
- •Statospores
- •Nutrition
- •Ecology
- •Classification
- •Chromulinales
- •Parmales
- •Chrysomeridales
- •REFERENCES
- •SYNUROPHYCEAE
- •Classification
- •REFERENCES
- •EUSTIGMATOPHYCEAE
- •REFERENCES
- •PINGUIOPHYCEAE
- •REFERENCES
- •DICTYOCHOPHYCEAE
- •Classification
- •Rhizochromulinales
- •Pedinellales
- •Dictyocales
- •REFERENCES
- •PELAGOPHYCEAE
- •REFERENCES
- •BOLIDOPHYCEAE
- •REFERENCE
- •BACILLARIOPHYCEAE
- •Cell structure
- •Cell wall
- •Cell division and the formation of the new wall
- •Extracellular mucilage, biolfouling, and gliding
- •Motility
- •Plastids and storage products
- •Resting spores and resting cells
- •Auxospores
- •Rhythmic phenomena
- •Physiology
- •Chemical defense against predation
- •Ecology
- •Marine environment
- •Freshwater environment
- •Fossil diatoms
- •Classification
- •Biddulphiales
- •Bacillariales
- •REFERENCES
- •RAPHIDOPHYCEAE
- •REFERENCES
- •XANTHOPHYCEAE
- •Cell structure
- •Cell wall
- •Chloroplasts and food reserves
- •Asexual reproduction
- •Sexual reproduction
- •Mischococcales
- •Tribonematales
- •Botrydiales
- •Vaucheriales
- •REFERENCES
- •PHAEOTHAMNIOPHYCEAE
- •REFERENCES
- •PHAEOPHYCEAE
- •Cell structure
- •Cell walls
- •Flagella and eyespot
- •Chloroplasts and photosynthesis
- •Phlorotannins and physodes
- •Life history
- •Classification
- •Dictyotales
- •Sphacelariales
- •Cutleriales
- •Desmarestiales
- •Ectocarpales
- •Laminariales
- •Fucales
- •REFERENCES
- •PRYMNESIOPHYCEAE
- •Cell structure
- •Flagella
- •Haptonema
- •Chloroplasts
- •Other cytoplasmic structures
- •Scales and coccoliths
- •Toxins
- •Classification
- •Prymnesiales
- •Pavlovales
- •REFERENCES
- •Toxic algae
- •Toxic algae and the end-Permian extinction
- •Cooling of the Earth, cloud condensation nuclei, and DMSP
- •Chemical defense mechanisms of algae
- •The Antarctic and Southern Ocean
- •The grand experiment
- •Antarctic lakes as a model for life on the planet Mars or Jupiter’s moon Europa
- •Ultraviolet radiation, the ozone hole, and sunscreens produced by algae
- •Hydrogen fuel cells and hydrogen gas production by algae
- •REFERENCES
- •Glossary
- •Index

378 CHLOROPLAST E.R.: EVOLUTION OF TWO MEMBRANES
Fig. 17.14 Scanning electron micrographs of silica spheres precipitated out of solution using two different silaffins and polyamines. The silica spheres in (a) are much smaller than the silica spheres in (b). The size of the silica spheres is controlled by the type of silaffin and/or polyamine in the silica deposition vesicle of the diatom. (From Kroger et al., 2000.)
(a)
(b)
(c)
(d)
Fig. 17.15 Drawings of the formation of the frustule of the centric diatom Coscinodiscus wailesii. Initially silica is deposited in the part of the frustule that will be next to the plasma membrane in the mature frustule (a) and (b). Subsequently the exterior portion of the frustule is deposited in (c) and (d). The mature frustule has a sieve membrane (s) on the exterior and a foramen (f) on the interior of the loculus. (From Schmid and Volcani, 1983.)
reducing the recreational suitability of the beaches. Extracellular mucilage production by diatoms is stimulated when phosphorus is limited, leading to marine snow blooms. Bacteria growing in the aggregates produce gas bubbles that give the marine snow positive buoyancy. The organisms in the aggregates benefit from reduced predation by grazers.
Diatoms are ubiquitous fouling microorganisms, attaching to submerged structures by secreting insoluble mucilages. Achnanthes longipes is a common marine fouling diatom that is highly resistant to toxic antifouling coatings (Johnson et al., 1995). It produces a stalk that elevates it above the toxic coatings on ship bottoms (Fig. 17.17). Fouling of ship bottoms increases the frictional drag, leading to excess fuel consumption. Cleaning ship hulls coasts millions of dollars each year, leading to an additional loss of revenue (Chiovitti et al., 2003).
Motility
Some diatoms are able to glide over the surface of a substrate, leaving a mucilaginous trail in their wake. Gliding is restricted to those pennate diatoms with a raphe (Figs. 17.3, 17.4, 17.5) and those centric diatoms with labiate processes (Figs. 17.19, 17.20, 17.32). The gliding movement is characterized by large fluctuations in the velocity of movement, with great changes in speed occurring within tenths of seconds (Edgar, 1979). In pennate diatoms, the path of the diatom seems to be essentially dependent on the shape of the raphe.
HETEROKONTOPHYTA, BACILLARIOPHYCEAE |
379 |
|
|
Fig. 17.16 Forms of extracellular mucilage in diatoms. (Modified from Hoagland et al., 1993.)
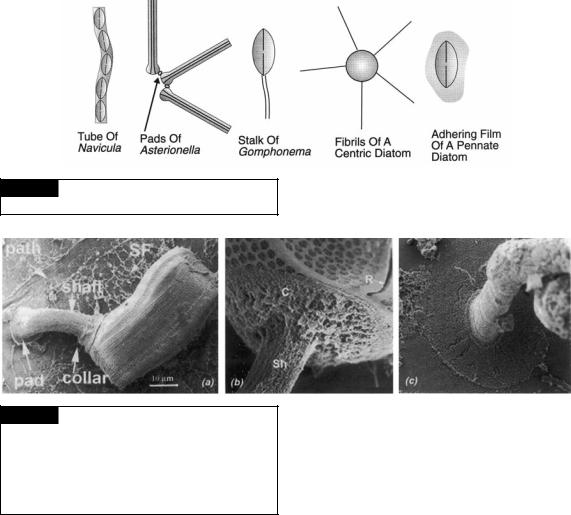
Fig. 17.17 Scanning electron micrographs of Achnanthes longipes. (a) Whole cell showing the pad, shaft, and collar of the mucilaginous stalk. Also shown is a path of mucilage left by the gliding diatom and surface film (SF) of mucilage left on the substratum. (b) The attachment of the collar (C) of the stalk (Sh) on the cell. (R) Raphe. (c) The attachment of the pad to the substratum. (From Wang et al., 2000.)
Nultsch (1956) distinguished at least three types:
(1) the Navicula type, with a straight movement; (2) the Amphora type, in which the path is usually curved; and (3) the Nitzschia type, which always exhibits curved pathways with two different radii. The observed rates of gliding in diatoms vary from 2 to 14 m s 1 at room temperature (Cohn and Weitzell, 1996). Nitzschia palea is able to penetrate into 2% agar and to move within the medium. The less solid the substrate is, the slower the movement of the diatom; the rate of movement of Nitzschia putrida was found to be 2.7 m s 1 on glass but only 0.8 m s 1 on agar (Wagner, 1934). Many of the diatoms exhibit backward and forward movements in which the direction may alternate at intervals of a minute. According to
von Denffer (1949), the motility of N. palea is dependent on light. In liquid cultures the cells tend to agglutinate into spherical clumps, loosely held together by mucilage. When they are transferred to a glass slide, the cells move away from one another. These movements do not take place in darkness.
Diatoms can glide only when the valve containing a raphe is in contact with the surface. If the diatom cell settles with the girdle contacting the substrate, the diatom secretes a mucilaginous tether from the portion of the raphe near the central nodule (Fig.17.18). The tether attaches to the substratum and the cell pulls itself onto a valve containing a raphe using the tether. Sometimes tethers are produced from raphes on opposite sides of the cell. This results in the cell being tethered on both sides of the cell. Withdrawal of the tethers causes the cell to rock back and forth until one of the tethers breaks and the cell rights itself on the substratum. The mucilage that comprises the tether is different from the mucilage involved in gliding (Higgins et al., 2003).

380 CHLOROPLAST E.R.: EVOLUTION OF TWO MEMBRANES
Fig. 17.18 Scanning electron micrographs of Pinnularia viridis.
(a) Cell showing raphe (longitudinal slit) in the middle of the valve (V). The girdle bands (GB) are located around the periphery of the cell. Arrowhead points to the central nodule. (b) A cell that has settled on the girdle bands. Tether mucilage is secreted from the raphes of each valve. The tether mucilage is attached to the substrate. (c) Tether mucilage extending from the raphe in the central nodule area to the substrate. (d) Tether mucilage connected to fine curved threads (arrowheads). (From Higgins et al., 2003.)
Those pennate diatoms that glide have bundles of actin microfilaments running parallel to the raphe (Pickett-Heaps et al., 1979a,b). The microfilament bundles may serve to orient crystalloid bodies containing mucilaginous material in the cytoplasm immediately below the raphe. On an appropriate stimulus, the mucilaginous material is released into the raphe system from the area of the central or terminal pore (Fig. 17.4) (Drum and Hopkins, 1966). The mucilaginous material then streams in the raphe in one direction until it strikes an object to which it adheres. If the object is fixed, then the streaming in the raphe forces the diatom to move in the opposite direction. Nearly all motile diatoms must adhere to the substratum in the area of their raphe in order for movement to occur.
Some of the benthic centric diatoms with labiate processes (Figs. 17.19, 17.20, 17.32) are capable of gliding when attached to a substrate. The labiate processes have a pore in the center, and the mucilage is secreted through the pore. In Actinocyclus subtilis (Fig. 17.20), secretion of the
mucilage through the labiate process causes the diatom to move forward while rotating at the same time (Medlin et al., 1986).
Diatoms that are attached to a substrate and are motile on the substrate have the advantages of
(1) being held in position in moving water; (2) avoiding burial by moving up and over sediments;
(3) moving to colonize vacant areas; and (4) moving to areas with more light and/or nutrients (Medlin et al., 1986).
Fig. 17.19 Drawing of a side view of Odontella sinensis showing the location of the labiate processes (L). (After Pickett-Heaps et al., 1986.)

HETEROKONTOPHYTA, BACILLARIOPHYCEAE |
381 |
|
|
Fig. 17.20 Scanning electron micrographs of Actinocyclus subtilis. (a) Whole cell. (b) Detail of the labiate processes, showing how they open to the outside by a simple hole. (c) Internal view of an acid-cleaned valve showing structure of two labiate processes. Internally, each labiate process consists of a short tube that projects from the valve mantle to a slit-like opening. Bar 1 m. (From Andersen et al., 1986.)
Plastids and storage products
The chloroplasts are surrounded by two membranes of the chloroplast envelope, outside of which are the two membranes of the chloroplast E.R., the outer membrane being continuous with the outer membrane of the nuclear envelope. The thylakoids within the chloroplast are grouped three to a band, and in most chloroplasts there is a more or less central pyrenoid (Fig. 17.1). The pyrenoid is usually crossed by widely spaced bands of thylakoids, which in some cases are reduced from the normal three thylakoids per band to two. The chloroplasts contain chlorophylls a, c1, and c2, with an a : c ratio of 4:1 in
Phaeodactylum tricornutum (Mann and Myers, 1968). Fucoxanthin is the principal carotenoid, giving the cells their golden-brown color. Fucoxanthin is an efficient carotenoid in the transfer of energy to chlorophyll a and is part of photosystem II of photosynthesis.
There are also colorless or apochlorotic diatoms that live on decaying marine vegetation and the mucilages of large seaweeds (Lewin and Lewin, 1967). With the light microscope these
diatoms have no apparent plastids, although when examined at the fine-structural level they can be seen to have proplastids with little if any internal membrane system. The proplastids have no detectable chlorophylls or carotenoids (Lauritis et al., 1968).
The storage product in the Bacillariophyceae is chrysolaminarin, which is located in vesicles in the cell (Granum and Myklestad, 2001). Chrysolaminarin differs from the laminarin found in the Phaeophyceae (Fig. 1.28) by lacking a terminal mannitol residue at the reducing end of the polysaccharide (Chiovitti et al., 2004).
Diatoms contain unique 4 -methyl sterols, such as 4-desmethylsterol and cholesterol (Fig. 17.21), that are useful as diatom markers in the water column and in organic sediments (Mansour et al., 1999).
Fig. 17.21 Two 4 -methyl sterols that occur in diatoms.
